 Back
BackChapter 9: Solutions – Key Concepts and Calculations
Study Guide - Smart Notes

Chapter 9: Solutions
9.1 Solutions: Definitions and Components
Solutions are homogeneous mixtures composed of two or more substances. The substance present in the largest amount is called the solvent, while the substance(s) present in lesser amounts are called solutes. In an aqueous solution, water acts as the solvent. Solutions are fundamental in chemistry and biology, as they allow for the uniform distribution of solutes at the molecular level.
Solution: Homogeneous mixture of two or more substances.
Solute: The component present in lesser quantity.
Solvent: The component present in the largest quantity.
Aqueous solution: A solution where water is the solvent.

Example: In a solution of salt water, salt is the solute and water is the solvent.
9.2 Solvation and Dissolution of Ionic Compounds
When an ionic compound such as sodium chloride (NaCl) dissolves in water, the process is called solvation. Water molecules surround and separate the ions from the crystal lattice, resulting in free-moving ions in solution. This process is essential for the conductivity of electrolytes in biological and chemical systems.
Solvation: The process of surrounding solute particles with solvent molecules.
Dissociation: Separation of an ionic compound into its constituent ions in solution.
Equation:
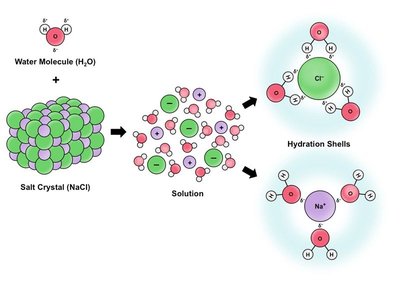
Example: When NaCl dissolves in water, each Na+ and Cl- ion is surrounded by water molecules, forming hydration shells.
9.2 Electrolytes and Non-electrolytes
Compounds that dissolve in water can be classified as electrolytes or non-electrolytes based on their ability to conduct electricity. Electrolytes dissociate into ions, enabling electrical conductivity, while non-electrolytes dissolve as neutral molecules and do not conduct electricity.
Electrolytes: Ionic compounds that dissociate into ions in water (e.g., NaCl, CaCl2).
Non-electrolytes: Covalent compounds that dissolve as molecules and do not conduct electricity (e.g., sugar, SO2).
Key distinction: Electrolytes are typically formed from metals and non-metals; non-electrolytes are formed from non-metals only.
Example: SO2 and P2O5 are non-electrolytes; CaCl2, LiNO3, and Na3PO4 are electrolytes.
9.3 Properties of Solutions
Solutions are characterized by their uniform appearance and properties. They are clear and transparent, with no visible particles, and do not scatter light. Solutions may be colored or colorless depending on the solute.
Homogeneity: Uniform composition throughout.
Transparency: No visible particles; does not scatter light.
Color: May be colored (e.g., copper sulfate solution) or colorless.
9.4 Concentration of Solutions: Molarity (M)
Molarity (M) is a common unit of concentration, defined as the number of moles of solute per liter of solution. It is used to quantify the amount of solute in a given volume of solution and is essential for preparing solutions and performing stoichiometric calculations.
Formula:
To find moles:
To find volume:
Always convert mL to L by dividing by 1000.
Example Calculation: Calculate the molarity of 2.0 L of solution containing 5.0 mol NaOH:
Example Calculation: A student prepares a 200 mL solution containing 0.30 mol HCl. What is the molarity?
Convert 200 mL to L:
Example Calculation: What is the concentration of a solution that contains 4.50 mol of NaF dissolved in 7.50 L of solution?
9.5 Calculating Moles from Molarity
To determine the number of moles of solute in a given volume of solution, multiply the molarity by the volume (in liters).
Formula:
Always convert mL to L before calculation.
Example: How many moles of NH3 are present in 1.50 L of a 0.500 M NH3 solution?
Example: Calculate the number of moles in 500 mL of 0.400 M HCl.
Convert 500 mL to L:
9.6 Applications of Solutions in Real Life
Solutions are essential in various real-life applications, including intravenous (IV) fluids in medicine, sports drinks for hydration, and laboratory preparations. The precise concentration of solutes in these solutions is critical for their effectiveness and safety.
Example: IV saline solutions must have the correct concentration of sodium chloride to match the osmolarity of blood.
