 Back
BackChapter 9: Solutions – Key Concepts and Applications
Study Guide - Smart Notes

Chapter 9: Solutions
9.1 Solutions
A solution is a homogeneous mixture composed of two or more substances. The substance present in the greatest amount is called the solvent, and the substance(s) present in lesser amounts are called solute(s). Solutions are important in both laboratory and real-world contexts, such as intravenous (IV) fluids used in medicine.
Example: Saline solution used in hospitals is a mixture of sodium chloride (NaCl) dissolved in water.
9.2 Electrolytes and Non-electrolytes
Electrolytes are substances that dissolve in water to produce ions, allowing the solution to conduct electricity. Non-electrolytes dissolve without forming ions and do not conduct electricity.
Electrolytes: Sodium chloride (NaCl), potassium chloride (KCl)
Non-electrolytes: Sugar (sucrose), ethanol
Application: Electrolyte solutions are critical in biological systems for nerve function and hydration.
9.3 Solubility
Solubility refers to the maximum amount of solute that can dissolve in a given amount of solvent at a specified temperature. The solubility of substances depends on temperature and, for gases, pressure.
Temperature: Most solids are more soluble at higher temperatures; gases are less soluble as temperature increases.
Pressure: Affects only gases; higher pressure increases gas solubility in liquids.
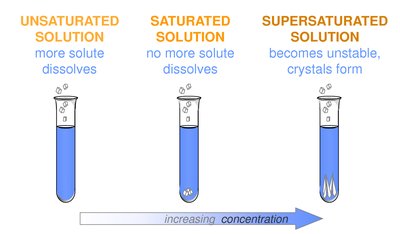
Types of Solutions:
Unsaturated: More solute can dissolve.
Saturated: No more solute dissolves; equilibrium is reached.
Supersaturated: Contains more solute than can normally dissolve; unstable and may form crystals.
Example: Sugar dissolving in water until no more can dissolve (saturated solution).
9.4 Concentration of Solutions
Concentration describes the amount of solute in a given quantity of solvent or solution. The most common unit is molarity (M), defined as moles of solute per liter of solution.
Molarity (M):

Example: A 0.10 M HCl solution contains 0.10 moles of HCl per liter.
9.5 Dilution of Solutions
Dilution is the process of adding solvent (usually water) to a solution to decrease its concentration. The amount of solute remains constant, but the volume increases, resulting in a lower molarity.
Key Principle: Moles of solute before and after dilution are equal.
Formula:

Example Calculation:
To dilute 0.050 L of 0.10 M HCl to 1.0 L: , ,
Find : M

Application: Dilution is used in preparing IV solutions and laboratory reagents.
9.6 Properties of Solutions
Solutions, colloids, and suspensions are types of mixtures with distinct properties. Their behavior depends on particle size and distribution.
Solution: Homogeneous, small particles (ions/molecules), do not settle, cannot be filtered.
Colloid: Larger particles, heterogeneous, do not settle, can be separated by semipermeable membranes.
Suspension: Very large particles, heterogeneous, settle rapidly, can be filtered.
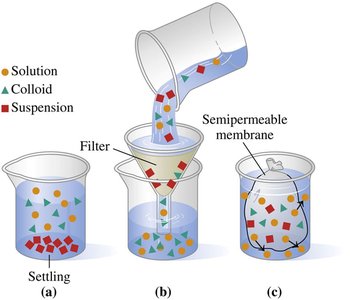
Examples:
Solution: Salt water, IV fluids
Colloid: Milk, mayonnaise

Suspension: Muddy water, salad dressing

Comparison Table: Solutions, Colloids, and Suspensions
Type of Mixture | Type of Particle | Settling | Separation |
|---|---|---|---|
Solution | Small particles (atoms, ions, molecules) | Do not settle | Cannot be separated by filters or semipermeable membranes |
Colloid | Larger molecules or groups of molecules/ions | Do not settle | Can be separated by semipermeable membranes, not by filters |
Suspension | Very large particles (may be visible) | Settle rapidly | Can be separated by filters |

Additional info: Colloids are important in biological systems (e.g., blood), and suspensions are common in environmental contexts (e.g., river water).
