 Back
BackChapter 9: Solutions – Properties, Calculations, and Types of Mixtures
Study Guide - Smart Notes

Chapter 9: Solutions
9.1 Solutions: Introduction and Definitions
Solutions are homogeneous mixtures composed of two or more substances. The substance present in the largest amount is called the solvent, while the other substances are solutes. Solutions are fundamental in chemistry due to their widespread occurrence and importance in both laboratory and real-world contexts.
Solvent: The component present in the greatest quantity; typically a liquid.
Solute: The component(s) present in lesser quantity; dissolved by the solvent.
Homogeneous mixture: Uniform composition throughout.
Examples: Salt water (NaCl in H2O), sugar in tea, air (gaseous solution).
9.2 Electrolytes and Non-electrolytes
When substances dissolve in water, they may or may not conduct electricity, depending on their ability to produce ions in solution. This property is used to classify solutions as electrolytes or non-electrolytes.
Electrolytes: Substances that dissolve in water to produce ions, allowing the solution to conduct electricity. Examples include ionic compounds like NaCl and KNO3.
Non-electrolytes: Substances that dissolve in water but do not produce ions; their solutions do not conduct electricity. Examples include most molecular compounds like CH4 (methane).
Example Classification:
Beaker 1: NO2 – weak electrolyte (partially ionizes)
Beaker 2: CH4 – non-electrolyte (does not ionize)
Beaker 3: KNO3 – strong electrolyte (fully ionizes)
9.3 Solubility
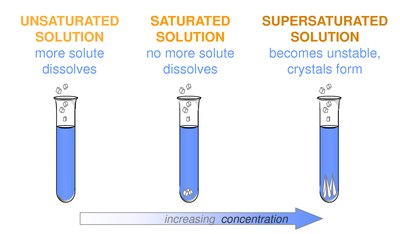
Solubility describes the maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature. Solutions can be classified based on their solute content relative to their solubility limit.
Unsaturated solution: Can dissolve more solute at the given temperature.
Saturated solution: Contains the maximum amount of dissolved solute; additional solute will not dissolve.
Supersaturated solution: Contains more solute than is normally possible at that temperature; unstable and may precipitate excess solute.
9.4 Concentration of Solutions: Molarity
The concentration of a solution is a measure of the amount of solute present in a given quantity of solvent or solution. Molarity (M) is the most common unit, defined as moles of solute per liter of solution.
Formula:
Calculating moles from molarity:
Volume from moles and molarity:
Unit conversions: 1 L = 1000 mL; always convert mL to L before calculations.
Example: What is the molarity of a solution made by dissolving 3.20 mol NaCl in 8.00 L water?
9.5 Dilution of Solutions
Dilution involves adding solvent to a solution, decreasing its concentration without changing the amount of solute. The relationship between the initial and final concentrations and volumes is given by the dilution equation.
Dilution equation:
= initial molarity
= initial volume
= final molarity
= final volume
Key point: The number of moles of solute remains constant during dilution.
Example: What is the molarity after diluting 0.050 L of 0.10 M HCl to 1.0 L?

9.6 Properties of Solutions
Solutions exhibit unique properties, such as uniform composition, inability to be separated by filtration, and stability over time. These properties distinguish solutions from other types of mixtures.
Homogeneous: Uniform distribution of solute particles.
Stable: Solute does not settle out over time.
Cannot be separated by filtration: Particles are too small to be trapped by filters.
Types of Liquid Mixtures: Solutions, Colloids, and Suspensions
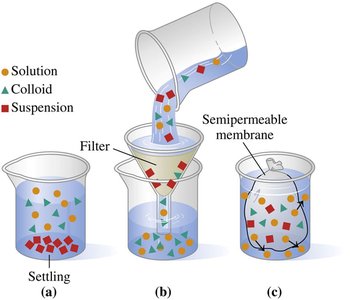
Not all mixtures are true solutions. Mixtures can be classified based on particle size and behavior as solutions, colloids, or suspensions.
Solution: Smallest particles (ions, atoms, small molecules); do not settle; cannot be separated by filters or membranes.
Colloid: Intermediate-sized particles; do not settle; can be separated by semipermeable membranes but not by filters. Examples: milk, mayonnaise.
Suspension: Largest particles; settle rapidly; can be separated by filters. Examples: muddy water, salad dressing.
Comparison Table: Solutions, Colloids, and Suspensions
Type of Mixture | Type of Particle | Settling | Separation |
|---|---|---|---|
Solution | Atoms, ions, small molecules | Do not settle | Cannot be separated by filters or membranes |
Colloid | Larger molecules or groups of molecules/ions | Do not settle | Can be separated by semipermeable membranes |
Suspension | Very large particles | Settle rapidly | Can be separated by filters |
Additional info: Understanding the differences between these types of mixtures is essential for laboratory techniques and for interpreting real-world phenomena such as blood analysis, water purification, and pharmaceutical preparations.
