 Back
BackChapter 9: Solutions – Properties, Types, and Calculations
Study Guide - Smart Notes

Solutions
Introduction to Solutions
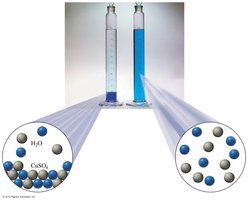
Solutions are homogeneous mixtures composed of two or more substances. They form when there is sufficient attraction between solute and solvent molecules. The solvent is present in a much larger amount, while the solute is present in a smaller amount.
Homogeneous mixture: Uniform composition throughout.
Solvent: Substance present in greater amount.
Solute: Substance present in lesser amount.

Example: Saltwater, where water is the solvent and salt is the solute.
Properties of Solutes
Solutes may be liquids, gases, or solids.
They are evenly spread throughout the solution.
Solutes mix with solvents to have the same physical state.
Cannot be separated by filtration but can be separated by evaporation.
Are not visible but can impart color to the solution.
Types of Solutes and Solvents
Solutes and solvents can be solids, liquids, or gases, leading to various types of solutions.
Type | Example | Primary Solute | Solvent |
|---|---|---|---|
Gas in gas | Air | Oxygen (gas) | Nitrogen (gas) |
Gas in liquid | Soda water | Carbon dioxide (gas) | Water (liquid) |
Liquid in liquid | Vinegar | Acetic acid (liquid) | Water (liquid) |
Solid in liquid | Seawater | Sodium chloride (solid) | Water (liquid) |
Solid in solid | Brass | Zinc (solid) | Copper (solid) |

Water as a Solvent
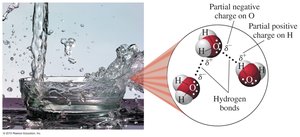
Water is the most common solvent in nature due to its polarity and ability to form hydrogen bonds. Its polar O—H bonds allow it to dissolve many ionic and polar substances.
Polar molecule: Uneven distribution of charge.
Hydrogen bonding: Strong intermolecular force important in biological systems.
Formation of Solutions: Like Dissolves Like
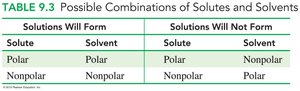
Solutions form when solute–solvent interactions are strong enough to overcome solute–solute and solvent–solvent interactions. The rule "like dissolves like" means polar solutes dissolve in polar solvents, and nonpolar solutes dissolve in nonpolar solvents.
Solutions Will Form | Solutions Will Not Form |
|---|---|
Polar solute + Polar solvent | Polar solute + Nonpolar solvent |
Nonpolar solute + Nonpolar solvent | Nonpolar solute + Polar solvent |
Solutions with Ionic and Polar Solutes
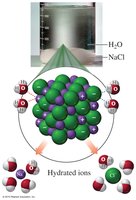
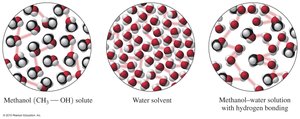
Ionic compounds like NaCl dissolve in water through hydration, where water molecules surround each ion. Polar molecular compounds, such as methanol, dissolve in water due to hydrogen bonding between the solute and solvent.
Hydration: Water molecules surround and separate ions.
Polar solutes: Require polar solvents for dissolution.
Electrolytes and Nonelectrolytes
Electrolytes in the Body
Electrolytes are substances that produce ions in solution and conduct electricity. They are essential for cellular and organ function. Examples include sodium, potassium, chloride, and bicarbonate.

Strong, Weak, and Nonelectrolytes
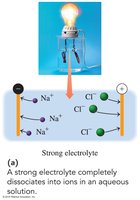
Strong electrolytes: Completely dissociate into ions in water, conducting electricity well.
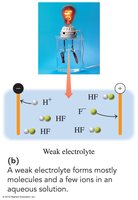
Weak electrolytes: Partially dissociate, producing few ions and weak conductivity.
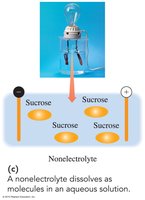
Nonelectrolytes: Dissolve as molecules, do not produce ions, and do not conduct electricity.
Type of Solute | In Solution | Conducts Electricity? | Examples |
|---|---|---|---|
Strong electrolyte | Dissociates completely | Yes | NaCl, KBr, HCl |
Weak electrolyte | Dissociates partially | Weakly | HF, H2O, NH3 |
Nonelectrolyte | No ionization | No | CH3OH, C12H22O11 |

Solubility
Solubility and Saturation
Solubility is the maximum amount of solute that dissolves in a specific amount of solvent at a given temperature. It is usually expressed as grams of solute per 100 grams of solvent.
Unsaturated solution: Contains less than the maximum solute; more can dissolve.
Saturated solution: Contains the maximum solute; excess remains undissolved.


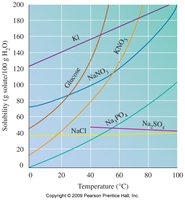
Effect of Temperature on Solubility
Solubility of most solids increases with temperature.
Solubility of gases decreases with temperature.
Concentration of Solutions
Units of Concentration
Concentration expresses the amount of solute in a given amount of solution. Common units include:
Mass percent (% m/m):
Volume percent (% v/v):
Mass/volume percent (% m/v):
Molarity (M):

CONCENTRATION: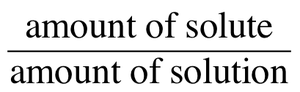
Calculating Mass Percent
To calculate mass percent, use the formula:
Volume Percent and Mass/Volume Percent
Volume percent is used for liquid solutes, and mass/volume percent is used for solid solutes in liquid solutions.
Molarity
Molarity (M) is the number of moles of solute per liter of solution:
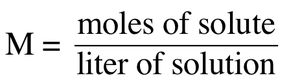


Dilution of Solutions
Principles of Dilution
Dilution involves adding solvent to a solution, increasing its volume and decreasing its concentration. The amount of solute remains constant.
For percent concentration:
For molarity:
Properties of Solutions: Solutions, Colloids, and Suspensions
Classification of Mixtures
Solutions: Transparent, do not separate, contain small particles that pass through filters and membranes.
Colloids: Medium-sized particles, cannot be filtered but can be separated by semipermeable membranes.
Suspensions: Heterogeneous, large particles that settle out and can be filtered.
Osmosis and Osmotic Pressure
Osmosis
Osmosis is the movement of water through a semipermeable membrane from a region of lower solute concentration to higher solute concentration. The process continues until equilibrium is reached.
Osmotic Pressure
Osmotic pressure is the pressure required to prevent the net flow of water into a solution. It increases with the number of dissolved particles.
M: Total particle molarity (osmolarity)
R: Gas constant
T: Temperature in Kelvin
Isotonic, Hypotonic, and Hypertonic Solutions
Isotonic: Same osmotic pressure as body fluids; cells retain normal volume.
Hypotonic: Lower solute concentration; water enters cells, causing swelling (hemolysis).
Hypertonic: Higher solute concentration; water leaves cells, causing shrinkage (crenation).
Dialysis
Dialysis is a process where small solute particles and solvent pass through an artificial membrane, while large particles are retained. It is used in medical treatments such as hemodialysis to remove waste from blood.
