 Back
BackChapter 9: Solutions – Structure, Properties, and Calculations
Study Guide - Smart Notes

Mixtures and Solutions
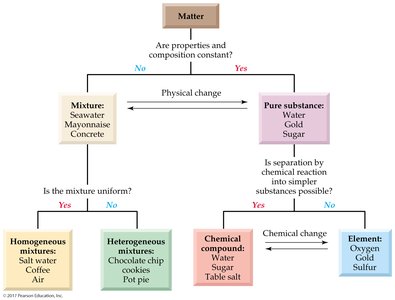
Classification of Matter
Matter can be classified based on the uniformity of its composition and the ability to separate its components by physical or chemical means. Understanding these classifications is fundamental to the study of chemistry and solutions.
Mixture: A combination of two or more substances where each retains its own properties. Mixtures can be separated by physical means.
Pure Substance: Has a constant composition and distinct chemical properties. Can be an element or a compound.
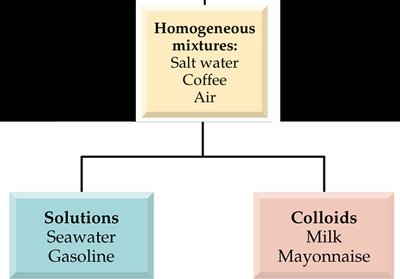
Homogeneous Mixture (Solution): Uniform composition throughout (e.g., salt water, air).
Heterogeneous Mixture: Non-uniform composition (e.g., chocolate chip cookies, pot pie).
Colloid: A mixture with intermediate particle size, often murky or opaque, does not separate on standing (e.g., milk, mayonnaise).
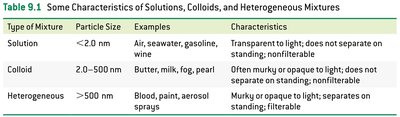
Characteristics of Solutions, Colloids, and Heterogeneous Mixtures
The properties of mixtures depend on the size of their particles and their behavior in light and filtration.
Type of Mixture | Particle Size | Examples | Characteristics |
|---|---|---|---|
Solution | < 2.0 nm | Air, seawater, gasoline, wine | Transparent to light; does not separate on standing; nonfilterable |
Colloid | 2.0–500 nm | Butter, milk, fog, pearl | Often murky or opaque to light; does not separate on standing; nonfilterable |
Heterogeneous | > 500 nm | Blood, paint, aerosol sprays | Murky or opaque to light; separates on standing; filterable |
Types of Solutions
Solutions can exist in various phases, depending on the physical states of the solute and solvent.
Type of Solution | Example |
|---|---|
Gas in gas | Air (O2, N2, Ar, other gases) |
Gas in liquid | Seltzer water (CO2 in water) |
Gas in solid | H2 in palladium metal |
Liquid in liquid | Gasoline (mixture of hydrocarbons) |
Liquid in solid | Dental amalgam (mercury in silver) |
Solid in liquid | Seawater (NaCl and other salts in water) |
Solid in solid | Metal alloys such as 14-karat gold (Au, Ag, Cu) |

Components of Solutions
Solute, Solvent, and Solution
A solution is a homogeneous mixture of two or more substances. The solvent is the substance present in the greatest amount, while the solute is present in a lesser amount. The solute dissolves in the solvent to form the solution.
Example: In salt water, water is the solvent and salt is the solute.

The Solution Process
How Solutions Form
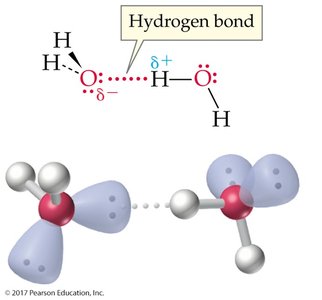
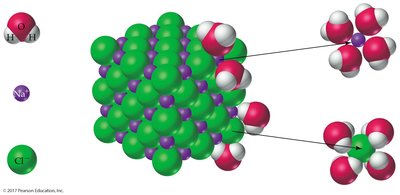
The process of dissolving involves interactions between solute and solvent particles. For ionic compounds, water molecules surround and separate the ions due to their polarity and ability to form hydrogen bonds.
"Like dissolves like": Polar solutes dissolve in polar solvents; nonpolar solutes dissolve in nonpolar solvents.
Example: NaCl dissolves in water due to ion-dipole interactions.
Solubility
Definition and Factors Affecting Solubility
Solubility is the maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature and pressure. Solubility depends on temperature, pressure (for gases), and the nature of the solute and solvent.
Saturated Solution: Contains the maximum amount of dissolved solute at equilibrium.
Unsaturated Solution: Contains less than the maximum amount of solute.
Supersaturated Solution: Contains more than the maximum amount of solute (unstable).

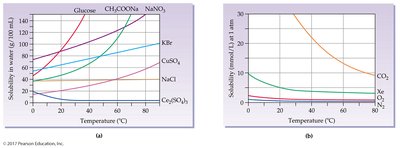
Effect of Temperature on Solubility
The solubility of most solids increases with temperature (endothermic dissolution), while the solubility of gases decreases with increasing temperature (exothermic dissolution).
Example: More sugar dissolves in hot tea than in cold tea.
Units of Concentration
Common Units
Concentration expresses the amount of solute in a given quantity of solution. Common units include:
Molarity (M):
Mass Percent (m/m%):
Mass/Volume Percent (m/v%):
Volume/Volume Percent (v/v%):
Example Calculation
Molarity: A solution contains 23 g of KI in 350 mL solution. Molarity =
Mass/Volume Percent:
Preparation and Dilution of Solutions
Preparing Solutions
To prepare a solution of known concentration from a solid solute:
Weigh the correct amount of solute.
Add to a volumetric flask.
Add some solvent and swirl to dissolve.
Fill to the calibration mark with solvent and mix thoroughly.

Dilution of Solutions
To dilute a concentrated solution to a lower concentration, use the formula:
M1: Initial molarity (concentrated solution)
V1: Volume of concentrated solution
M2: Final molarity (diluted solution)
V2: Final total volume
Ions in Solution: Electrolytes
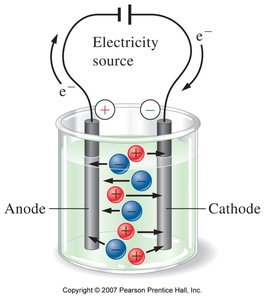
Electrolytes and Conductivity
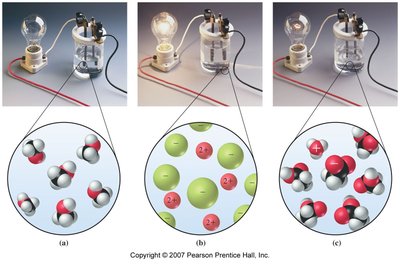
Electrolytes are substances that conduct electricity when dissolved in water due to the presence of ions.
Strong Electrolytes: Dissociate completely (e.g., NaCl, HCl).
Weak Electrolytes: Partially dissociate (e.g., acetic acid).
Non-electrolytes: Do not produce ions (e.g., glucose).

Chemical Equations for Dissolution
Ionic Compound:
Strong Acid:
Weak Acid:
Properties of Solutions (Colligative Properties)
Boiling Point Elevation and Freezing Point Depression
The presence of solute particles affects the physical properties of solutions:
Boiling Point Elevation: Solutions boil at higher temperatures than pure solvents.
Freezing Point Depression: Solutions freeze at lower temperatures than pure solvents.
Vapor Pressure Lowering: Solutions have lower vapor pressure than pure solvents.
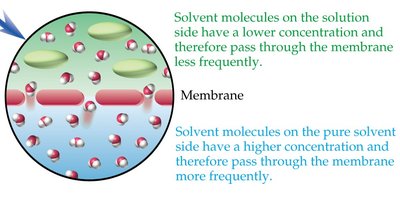
Osmosis and Osmotic Pressure
Osmosis is the movement of solvent molecules through a semipermeable membrane from a region of lower solute concentration to higher solute concentration. Osmotic pressure is the pressure required to stop this flow.

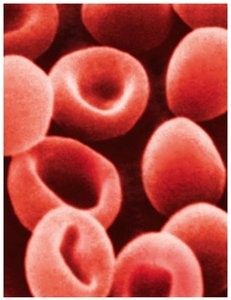
Osmotic Effects on Cells
Isotonic Solution: Same osmotic pressure as cells; no net water movement.
Hypotonic Solution: Lower osmotic pressure; water enters cells, causing swelling (hemolysis).
Hypertonic Solution: Higher osmotic pressure; water leaves cells, causing shrinkage (crenation).
Summary and Study Tips
Review the definitions and properties of mixtures, solutions, and colloids.
Practice calculations involving concentration units and dilution.
Understand the effects of solutes on boiling point, freezing point, and osmotic pressure.
Apply concepts to real-world examples, such as medical solutions and environmental chemistry.
