 Back
BackCHEM 105 Test 2 Study Guide – Step-by-Step Guidance
Study Guide - Smart Notes

Q1. Fill out this periodic table with the names of groups of elements: Alkali metals, alkaline earth metals, transition metals, halogens, noble gases, actinides, lanthanides.
Background
Topic: Periodic Table Organization
This question tests your understanding of the major groups and families of elements in the periodic table, which is foundational for predicting element properties and chemical behavior.
Key Terms:
Alkali metals: Group 1 elements (except hydrogen)
Alkaline earth metals: Group 2 elements
Transition metals: Groups 3–12
Halogens: Group 17 elements
Noble gases: Group 18 elements
Actinides: Bottom row of the two separated rows
Lanthanoids: Top row of the two separated rows
Step-by-Step Guidance
Review the periodic table layout and identify the location of each group listed above.
Label Group 1 (first column, except hydrogen) as "Alkali metals." Group 2 (second column) as "Alkaline earth metals."
Mark Groups 3–12 (middle block) as "Transition metals."
Label Group 17 (second-to-last column) as "Halogens," and Group 18 (last column) as "Noble gases."
Identify the two rows below the main table: the top row is "Lanthanides," and the bottom row is "Actinides."

Try solving on your own before revealing the answer!
Final Answer:
The periodic table should be labeled as follows: Group 1 – Alkali metals, Group 2 – Alkaline earth metals, Groups 3–12 – Transition metals, Group 17 – Halogens, Group 18 – Noble gases, top separated row – Lanthanides, bottom separated row – Actinides.
These group names help organize the periodic table and predict element properties.
Q3. Write the atomic symbol for the following atoms given their protons, neutrons, and electrons:
Background
Topic: Atomic Structure and Notation
This question tests your ability to use the number of protons, neutrons, and electrons to write the correct atomic symbol, including charge and mass number.
Key Terms and Formula:
Atomic number (): Number of protons
Mass number (): (protons + neutrons)
Charge:
Atomic symbol format:

Step-by-Step Guidance
For each atom, identify the number of protons (), neutrons (), and electrons.
Calculate the mass number () for each atom: .
Determine the element symbol using the atomic number () from the periodic table.
Calculate the charge: .
Write the atomic symbol in the format for each atom.

Try solving on your own before revealing the answer!
Final Answer:
Each atom's symbol is written as , where is the mass number, is the atomic number, and the charge is calculated from protons and electrons.
For example, Atom 1: , Atom 2: , etc.
Q7. Calculate the energy required for the following heating/cooling scenarios:
Background
Topic: Heat and Energy Calculations
This question tests your ability to use the specific heat formula to calculate the energy required to change the temperature of a substance.
Key Formula:
= heat energy (Joules)
= mass (grams or kilograms)
= specific heat capacity (J/g°C or J/kg°C)
= change in temperature ()

Step-by-Step Guidance
Identify the mass (), specific heat (), and initial and final temperatures for each scenario.
Calculate for each: .
Find the specific heat value for the substance from the provided table.
Plug the values into the formula for each scenario.
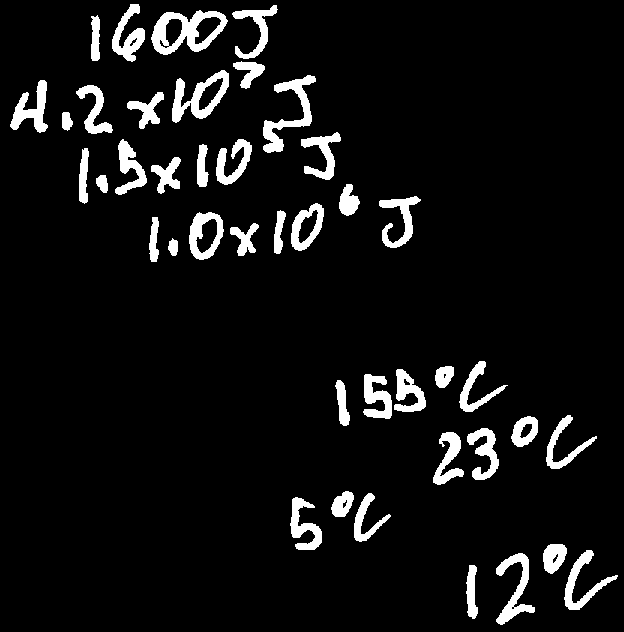
Try solving on your own before revealing the answer!
Final Answer:
For each scenario, the energy required is calculated using with the appropriate values for mass, specific heat, and temperature change.
For example, heating 15g of aluminum from 23°C to 145°C: .
Q9. Sketch the shape of the following orbitals: S-orbital, P-orbital, D-orbital, F-orbital.
Background
Topic: Atomic Orbitals and Quantum Chemistry
This question tests your understanding of the spatial shapes of atomic orbitals, which are important for predicting chemical bonding and electron distribution.
Key Terms:
S-orbital: Spherical shape
P-orbital: Dumbbell shape (two lobes)
D-orbital: Cloverleaf shape (four lobes or donut)
F-orbital: Complex, multi-lobed shape
Step-by-Step Guidance
Recall the basic shapes: S (sphere), P (dumbbell), D (cloverleaf), F (complex).
Draw each orbital shape, labeling the axes and nodes if needed.
Compare the number of lobes and symmetry for each orbital type.
Try sketching these shapes on your own before revealing the answer!
Final Answer:
S-orbital: Sphere; P-orbital: Dumbbell; D-orbital: Cloverleaf; F-orbital: Multi-lobed complex shape.
These shapes reflect the probability distributions of electrons in each orbital type.
Q12. Rank each of the following sets of elements from smallest to largest based on atomic size:
Background
Topic: Periodic Trends – Atomic Radius
This question tests your understanding of how atomic size changes across periods and groups in the periodic table.
Key Concepts:
Atomic size increases down a group and decreases across a period (left to right).
Compare elements in the same group or period to determine relative sizes.

Step-by-Step Guidance
Identify the position of each element in the periodic table.
Recall the trend: atomic radius increases down a group and decreases across a period.
Arrange the elements in each set from smallest to largest based on their group and period positions.
Try ranking the elements before revealing the answer!
Final Answer:
Each set is ranked from smallest to largest atomic size according to periodic trends. For example, Li < Na < K < Rb.
Atomic size increases as you move down a group and decreases across a period.
Q13. Rank each of the following sets of elements from smallest to largest based on ionization energy:
Background
Topic: Periodic Trends – Ionization Energy
This question tests your understanding of how ionization energy changes across periods and groups in the periodic table.
Key Concepts:
Ionization energy increases across a period (left to right) and decreases down a group.
Elements with higher ionization energy are harder to remove electrons from.
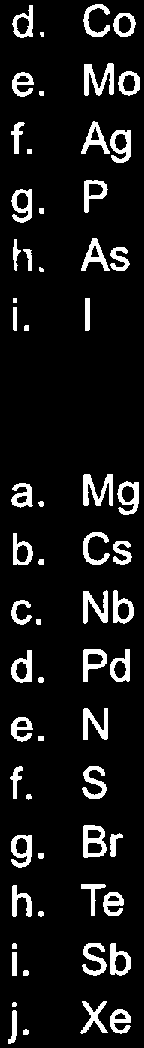
Step-by-Step Guidance
Identify the position of each element in the periodic table.
Recall the trend: ionization energy increases across a period and decreases down a group.
Arrange the elements in each set from smallest to largest based on their group and period positions.
Try ranking the elements before revealing the answer!
Final Answer:
Each set is ranked from smallest to largest ionization energy according to periodic trends. For example, Rb < K < Na < Li.
Ionization energy increases as you move across a period and decreases down a group.
