 Back
BackChem121 Midterm #2 Study Guide – Step-by-Step Guidance
Study Guide - Smart Notes

Q1. Write the formula for each of the following molecular compounds:
sulfur dioxide
dinitrogen trioxide
boron trifluoride
nitrogen dioxide
Background
Topic: Naming and Writing Formulas for Binary Covalent Compounds
This question tests your ability to convert the names of binary covalent (molecular) compounds into their correct chemical formulas using prefixes and element symbols.
Key Terms and Concepts:
Binary covalent compound: A compound composed of two different nonmetal elements.
Prefixes: Indicate the number of atoms of each element (e.g., mono-, di-, tri-, tetra-, penta-, etc.).
Element symbols: Use the periodic table to find the correct symbols for each element.
Step-by-Step Guidance
Identify the elements present in each compound name and write their symbols in the order given.
Use the prefixes to determine the number of atoms of each element. For example, 'di-' means 2, 'tri-' means 3, 'tetra-' means 4, etc.
If there is no prefix for the first element, it is assumed to be one atom (do not write a subscript of 1).
Write the formula by placing the appropriate subscript after each element symbol to indicate the number of atoms.

Try solving on your own before revealing the answer!
Q2. Arrange the following bonds in order of most polar to least polar using electronegativity values:
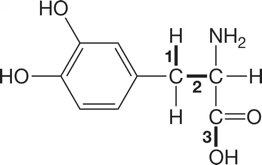
Background
Topic: Bond Polarity and Electronegativity
This question asks you to use the concept of electronegativity to determine the relative polarity of different bonds in a molecule. The greater the difference in electronegativity between two bonded atoms, the more polar the bond.
Key Terms and Concepts:
Electronegativity: A measure of an atom's ability to attract shared electrons in a chemical bond.
Bond polarity: The unequal sharing of electrons between two atoms due to differences in electronegativity.
Most polar bond: The bond with the largest electronegativity difference.
Step-by-Step Guidance
Identify the atoms involved in each numbered bond in the structure.
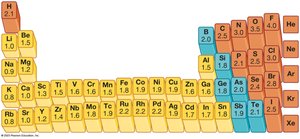
Look up the electronegativity values for each atom using a periodic table with electronegativity values.
Calculate the difference in electronegativity for each bond.
Rank the bonds from largest to smallest electronegativity difference (most polar to least polar).
Try solving on your own before revealing the answer!
Q3. Draw the Lewis structure for each molecule, including lone pairs of electrons:
PBr3
CH2O
Background
Topic: Lewis Structures for Covalent Compounds
This question tests your ability to draw Lewis structures, which show how valence electrons are arranged among atoms in a molecule, including bonding pairs and lone pairs.
Key Terms and Concepts:
Lewis structure: A diagram showing the arrangement of valence electrons around atoms in a molecule.
Lone pairs: Pairs of valence electrons not involved in bonding.
Bonding pairs: Electrons shared between atoms to form covalent bonds.
Step-by-Step Guidance
Count the total number of valence electrons for all atoms in the molecule.
Arrange the atoms with the least electronegative atom (except hydrogen) in the center.
Connect the atoms with single bonds, then distribute remaining electrons as lone pairs to satisfy the octet rule (or duet for hydrogen).
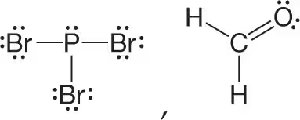
Try solving on your own before revealing the answer!
