 Back
BackChemical Quantities and Chemical Reactions: Study Notes
Study Guide - Smart Notes

Chemical Quantities and the Mole
Understanding the Mole Concept
The mole is a fundamental unit in chemistry that allows chemists to count atoms, molecules, or ions by weighing them. It is analogous to familiar counting units such as a dozen or a gross.
Mole (mol): The amount of substance that contains as many particles (atoms, molecules, ions) as there are atoms in exactly 12 grams of carbon-12.
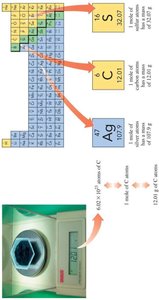
Avogadro's Number: $6.022 \times 10^{23}$ particles per mole.
Counting Units: Just as 1 dozen = 12 items, 1 mole = $6.022 \times 10^{23}$ particles.
Example: One mole of sulfur contains $6.02 \times 10^{23}$ sulfur atoms.


Avogadro's Number and Conversions
Avogadro's number is used to convert between the number of particles and moles of a substance.
Conversion: Number of particles $\leftrightarrow$ Moles using Avogadro's number.
Formula: $\text{Number of particles} = \text{moles} \times 6.022 \times 10^{23}$

Molecular Compounds and Moles
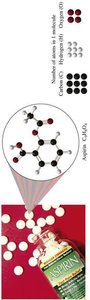
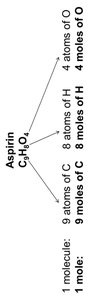
The mole concept applies to molecules as well as atoms. For example, aspirin (C9H8O4) contains a specific number of each type of atom per molecule and per mole.
Example: 1 molecule of aspirin contains 9 C, 8 H, and 4 O atoms; 1 mole contains 9 moles C, 8 moles H, and 4 moles O.
Molar Mass and Chemical Calculations
Calculating Molar Mass
The molar mass of a substance is the mass in grams of one mole of that substance. It is numerically equal to the atomic or molecular mass in atomic mass units (amu), but expressed in grams per mole (g/mol).
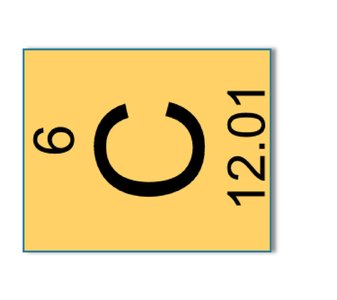
Atomic Mass: The mass of one mole of an element (e.g., Na: 22.99 g/mol, C: 12.01 g/mol).
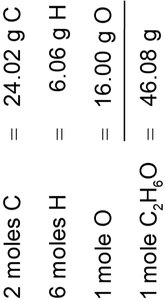
Molar Mass of Compounds: Sum the molar masses of all atoms in the formula.
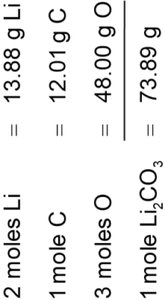
Example: Molar mass of Li2CO3 = 2(6.94) + 12.01 + 3(16.00) = 73.89 g/mol.


Using Molar Mass as a Conversion Factor
Molar mass is used to convert between grams and moles of a substance.
Formula: $\text{Moles} = \frac{\text{Mass (g)}}{\text{Molar Mass (g/mol)}}$
Formula: $\text{Mass (g)} = \text{Moles} \times \text{Molar Mass (g/mol)}$


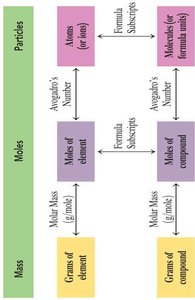
Summary Table: Relationships Among Mass, Moles, and Particles
This table summarizes the relationships and conversion factors among mass, moles, and particles for elements and compounds.
Mass | Moles | Particles |
|---|---|---|
Grams of element/compound | Moles of element/compound | Atoms, molecules, or formula units |
Molar Mass (g/mol) | Avogadro's Number | Formula Subscripts |
Chemical Reactions and Equations
Evidence of Chemical Change
Chemical reactions involve the transformation of substances into new products. Evidence includes color change, formation of a precipitate, gas production, or energy change.
Example: Tarnishing of silver (Ag) forms silver sulfide (Ag2S).
Example: Rusting of iron (Fe) forms iron(III) oxide (Fe2O3).


Writing and Balancing Chemical Equations
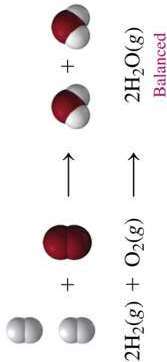
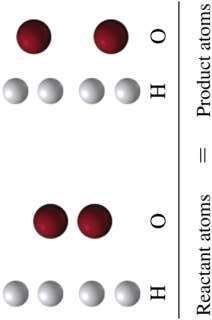
Chemical equations represent the reactants and products in a reaction. Balancing ensures the law of conservation of mass is obeyed.
Example: $\text{C (s) + O}_2\text{(g)} \rightarrow \text{CO}_2\text{(g)}$
Balanced Equation: $2\text{H}_2\text{(g)} + \text{O}_2\text{(g)} \rightarrow 2\text{H}_2\text{O(g)}$

Types of Chemical Reactions
Chemical reactions are classified into several types based on the rearrangement of atoms and molecules.
Combination (Synthesis): Two or more reactants combine to form a single product. $A + B \rightarrow AB$
Decomposition: A single reactant splits into two or more products. $AB \rightarrow A + B$
Single Replacement: One element replaces another in a compound. $A + BC \rightarrow AC + B$
Double Replacement: Two elements in different compounds exchange places. $AB + CD \rightarrow AD + CB$



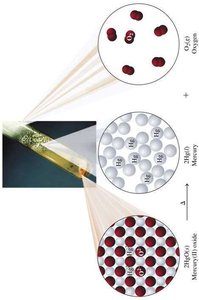
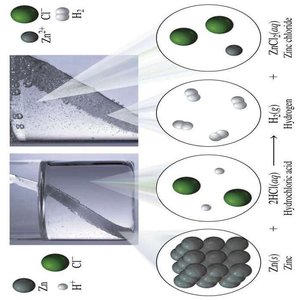
Redox (Oxidation-Reduction) Reactions
Redox reactions involve the transfer of electrons between substances. Oxidation is the loss of electrons, and reduction is the gain of electrons.
Oxidation: Loss of electrons (increase in oxidation state).
Reduction: Gain of electrons (decrease in oxidation state).
Example: Rusting of iron, tarnishing of silver, and combustion are all redox reactions.

Energy Changes in Chemical Reactions
Exothermic and Endothermic Reactions
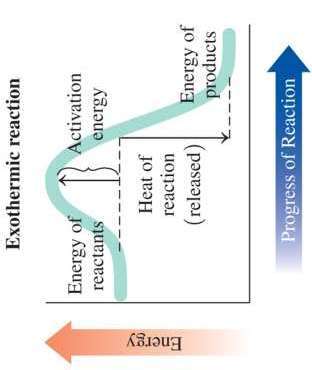
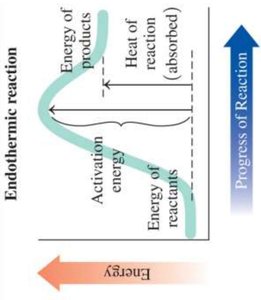
Chemical reactions involve energy changes. Exothermic reactions release energy, while endothermic reactions absorb energy.
Exothermic: Energy of products is lower than reactants; heat is released. Example: Combustion.
Endothermic: Energy of products is higher than reactants; heat is absorbed. Example: Photosynthesis.

Catalysts and Activation Energy
Catalysts lower the activation energy required for a reaction, increasing the reaction rate without being consumed.
Activation Energy: The minimum energy required for a reaction to occur.
Catalyst: Substance that increases reaction rate by providing an alternative pathway with lower activation energy.
Summary Table: Types of Chemical Reactions
Type | General Equation | Description |
|---|---|---|
Combination | $A + B \rightarrow AB$ | Two or more reactants form one product |
Decomposition | $AB \rightarrow A + B$ | One reactant splits into two or more products |
Single Replacement | $A + BC \rightarrow AC + B$ | One element replaces another in a compound |
Double Replacement | $AB + CD \rightarrow AD + CB$ | Two elements in different compounds exchange places |
Combustion | Hydrocarbon + O2 $\rightarrow$ CO2 + H2O | Reaction with oxygen producing heat and light |
