Back
BackChemical Quantities and Reactions: Study Guide
Study Guide - Smart Notes

CHAPTER 7: CHEMICAL QUANTITIES & REACTIONS
The Mole and Avogadro’s Number
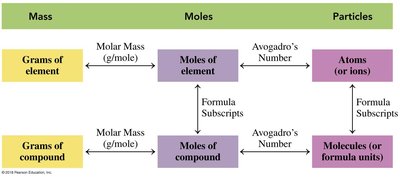
The concept of the mole is fundamental in chemistry for quantifying substances. One mole is defined as exactly 6.02 × 1023 particles (atoms, molecules, or formula units), known as Avogadro’s Number. The mole allows chemists to relate mass, number of particles, and volume in chemical calculations.
Mole: A counting unit, similar to a dozen, but for atoms/molecules.
Avogadro’s Number: particles per mole.
Application: Used to convert between mass, number of particles, and moles.

Moles per Element in a Compound
Each compound contains a specific number of moles of each element, determined by its chemical formula. For example, in NaCl, 1 mole contains 1 mole of Na and 1 mole of Cl. In more complex compounds, the mole ratios reflect the subscripts in the formula.
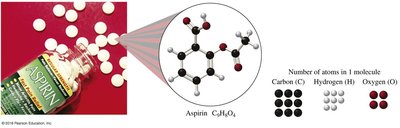
Example: Aspirin (C9H8O4) contains 9 moles of C, 8 moles of H, and 4 moles of O per mole of aspirin.
Formula Subscripts: Indicate the number of moles of each element per mole of compound.
Molar Mass
The molar mass is the mass of one mole of a substance, expressed in grams per mole (g/mol). It is calculated by summing the atomic masses of all atoms in a chemical formula.
Calculation: Add atomic masses from the periodic table for each element in the formula.
Example: Molar mass of NaCl = mass of Na + mass of Cl.
Use: Converts between grams and moles.
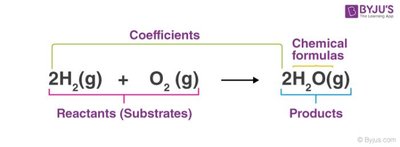
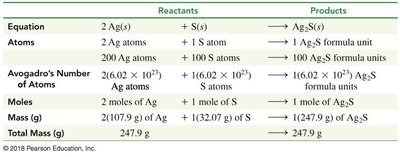
Chemical Equations and Balancing
Chemical equations represent reactions, showing reactants and products. Balancing equations ensures the law of conservation of mass is obeyed: the number of atoms of each element must be equal on both sides.
Reactants: Substances consumed in the reaction.
Products: Substances formed in the reaction.
Coefficients: Numbers placed before formulas to balance atoms.
Balancing Steps: Adjust coefficients, not subscripts; balance polyatomic ions as units.


Types of Chemical Reactions
Chemical reactions are classified by the changes occurring in reactants and products. The main types are combination, decomposition, single replacement, double replacement, and combustion.
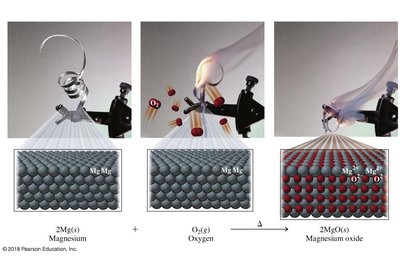
Combination: Two or more reactants form one product.
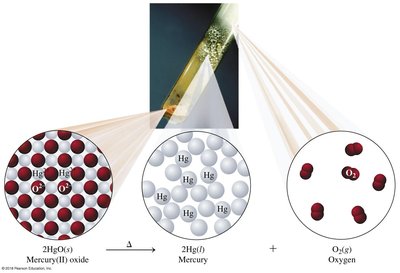
Decomposition: One reactant splits into two or more products.
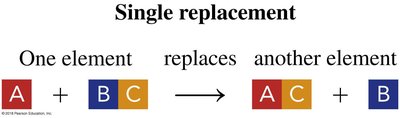
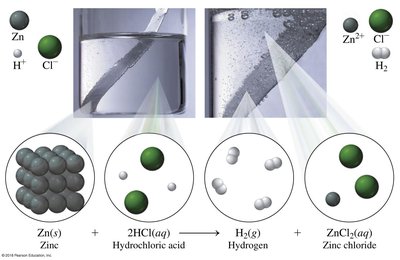
Single Replacement: One element replaces another in a compound.
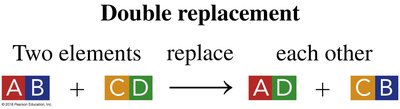
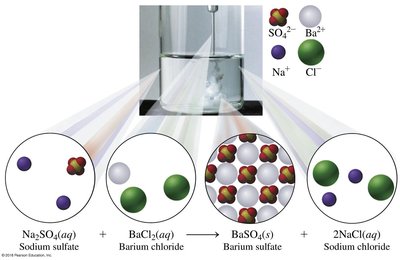
Double Replacement: Two compounds exchange elements to form new compounds.
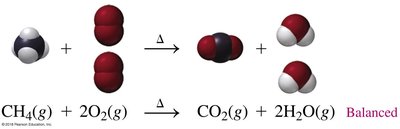
Combustion: Fuel reacts with oxygen to produce CO2, H2O, and energy.


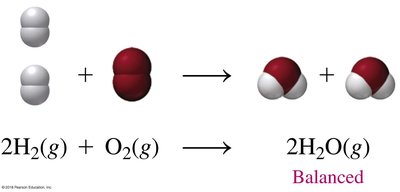



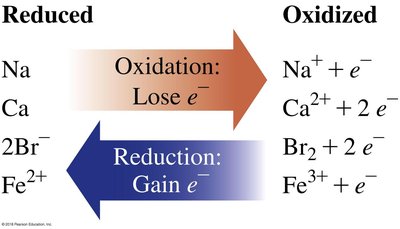
Oxidation and Reduction (Redox Reactions)
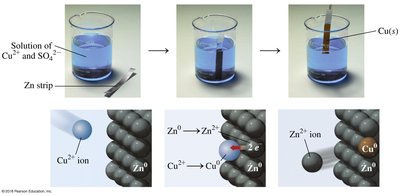
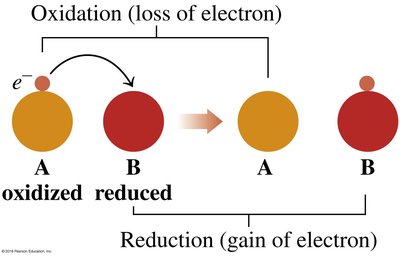
Redox reactions involve the transfer of electrons. Oxidation is the loss of electrons, while reduction is the gain of electrons. The mnemonic "OIL RIG" helps remember: Oxidation Is Losing, Reduction Is Gaining (electrons).
Oxidation: Element loses electrons, increases oxidation state.
Reduction: Element gains electrons, decreases oxidation state.
Example: Mg metal oxidized to Mg2+; O2 reduced to O2−.
Summary of Reaction Types
The following table summarizes the main types of reactions and provides examples for each.
Reaction Type | Example |
|---|---|
Combination | Ca(s) + Cl2(g) → CaCl2(s) |
Decomposition | FeS2(s) → 2Fe(s) + 3S(s) |
Single Replacement | Cu(s) + 2AgNO3(aq) → 2Ag(s) + Cu(NO3)2(aq) |
Double Replacement | BaCl2(aq) + K2SO4(aq) → BaSO4(s) + 2KCl(aq) |
Combustion | CH4(g) + 2O2(g) → CO2(g) + 2H2O(g) + energy |

Mole-Mole Factors in Reactions
Balanced chemical equations provide mole-mole factors, which are ratios used to convert between moles of different substances in a reaction.
Example: In 2H2 + O2 → 2H2O, the ratio is 2 mol H2 : 1 mol O2 : 2 mol H2O.
Use: Essential for stoichiometric calculations.
Mass Calculations in Reactions (Stoichiometry)
Stoichiometry involves calculating the mass of reactants and products using balanced equations, molar masses, and mole-mole factors.
Steps:
Convert grams to moles using molar mass.
Use mole-mole factor from balanced equation.
Convert moles to grams using molar mass.
Example: Calculate mass of CO2 produced from a given mass of C2H2.



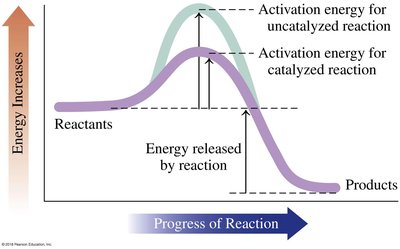
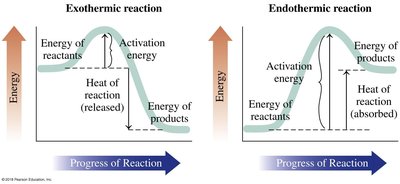
Exothermic vs Endothermic Reactions
Chemical reactions can either release energy (exothermic) or absorb energy (endothermic). The energy profile of a reaction shows the activation energy and the overall energy change.
Exothermic: Energy released; products have lower energy than reactants.
Endothermic: Energy absorbed; products have higher energy than reactants.
Activation Energy: Minimum energy required to initiate a reaction.
Reaction Rate (Kinetics)
The rate of a reaction describes how quickly reactants are converted to products. Factors affecting rate include concentration, temperature, and presence of a catalyst.
Catalyst: Lowers activation energy, increasing reaction rate.
Energy Profile: Shows difference in activation energy with and without catalyst.