 Back
BackChemical Quantities and Reactions: Study Guide
Study Guide - Smart Notes

Chemical Quantities and Reactions
The Mole Concept and Atoms
The mole is a fundamental unit in chemistry used to express amounts of a chemical substance. One mole contains Avogadro's number of particles (6.022 x 1023), whether they are atoms, molecules, or ions.
Mole (mol): The SI unit for amount of substance.
Avogadro's Number: 6.022 x 1023 particles per mole.
Application: Used to relate mass, number of particles, and volume in chemical calculations.
Chemical Formula, Formula Mass, and Molar Mass
Chemical formulas represent the composition of compounds. The formula mass is the sum of the atomic masses of all atoms in a formula unit, and the molar mass is the mass of one mole of a substance, expressed in grams per mole (g/mol).
Chemical Formula: Shows the types and numbers of atoms in a compound (e.g., Al(OH)3).
Formula Mass: Sum of atomic masses in a formula unit.
Molar Mass: Mass of one mole of a substance (g/mol).
Example: Molar mass of aluminum hydroxide, Al(OH)3 = 78.01 g/mol.
Example: Molar mass of lithium carbonate, Li2CO3 = 73.89 g/mol.

Mass to Mole Conversions
Mass-to-mole conversions are essential for quantifying substances in chemical reactions. The molar mass serves as a conversion factor between grams and moles.
Conversion Formula:
Reverse Conversion:
Example: 125 g of NH4Cl (molar mass 53.50 g/mol) contains 2.34 mol.
Example: 2.50 mol of C2H6 (molar mass 30.08 g/mol) has a mass of 75.2 g.
Example: 737 g of NaCl (molar mass 58.44 g/mol) contains 12.6 mol.
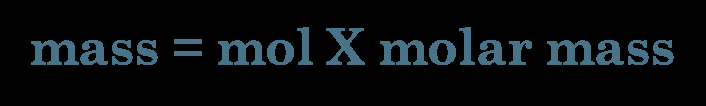
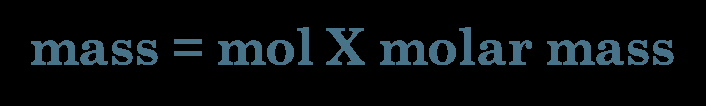
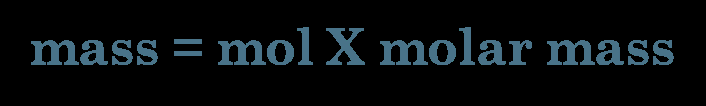
Chemical Equations
Chemical equations are shorthand notations for chemical reactions, showing reactants, products, physical states, and conditions. They must be balanced to obey the law of conservation of mass.
Reactants: Substances that undergo change (left side of the arrow).
Products: Substances produced (right side of the arrow).
Physical States: Indicated by (s) for solid, (l) for liquid, (g) for gas, (aq) for aqueous.
Energy: Symbol ∆ over the arrow means energy is required.
Balanced Equation: Number of atoms of each element must be equal on both sides.
Example: 2HgO(s) 2Hg(l) + O2(g)
Evidence of a Reaction Occurring
Chemical reactions can be identified by observable changes.
Release of a gas
Formation of a solid (precipitate)
Heat produced or absorbed
Color changes

Classification of Chemical Reactions
Chemical reactions are classified by their patterns, which helps in predicting products and understanding reaction mechanisms.
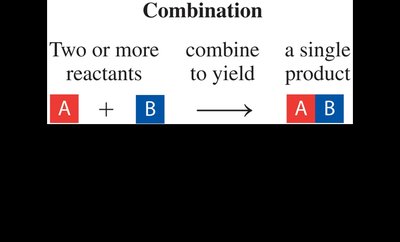
Combination Reaction: Two or more reactants combine to form a single product.
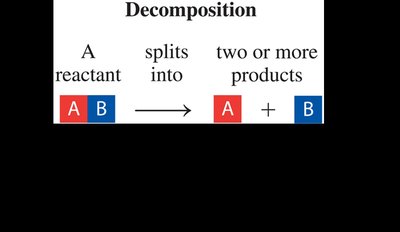
Decomposition Reaction: A single reactant splits into two or more products.
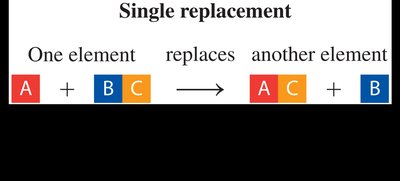
Single-Replacement Reaction: One element replaces another in a compound.
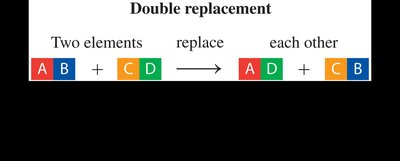
Double-Replacement Reaction: Two elements in different compounds exchange places.
Sample Calculations and Practice Problems
Practice problems reinforce understanding of molar mass calculations and mass-mole conversions.
Calculate the molar mass of glucose, C6H12O6: 180.18 g/mol
Calculate the mass for 0.100 mol of glucose:
Calculate moles from mass:
Compound | Molar Mass (g/mol) |
|---|---|
Al(OH)3 | 78.01 |
Li2CO3 | 73.89 |
NH4Cl | 53.50 |
NaCl | 58.44 |
C2H6 | 30.08 |
C6H12O6 | 180.18 |
LiCl | 42.39 |
Example: A student weighs 0.550 g of LiCl. Moles = 0.550 g / 42.39 g/mol = 0.0130 mol.
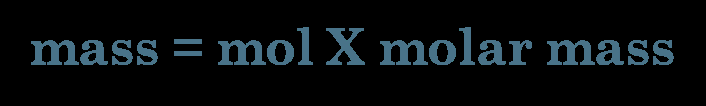
Additional info: Images of antacid and lithium carbonate medication reinforce real-world applications of molar mass calculations in pharmaceuticals and consumer products.
