 Back
BackChemical Reactions and Balancing Equations – Study Notes
Study Guide - Smart Notes

Chemical Reactions and Their Classification
Types of Chemical Reactions
Chemical reactions can be classified into several fundamental types based on the rearrangement of atoms and the nature of the reactants and products. Understanding these types is essential for predicting reaction outcomes and balancing equations.
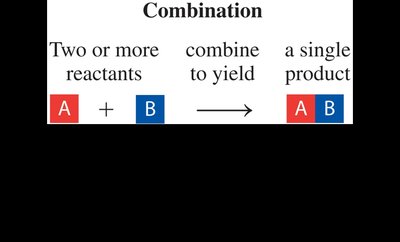
Combination (Synthesis) Reaction: Two or more reactants combine to form a single product.
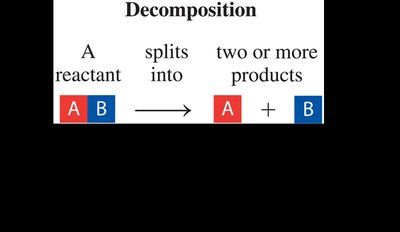
Decomposition Reaction: A single compound breaks down into two or more simpler substances.
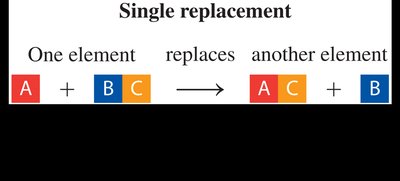
Single Replacement Reaction: One element replaces another in a compound.
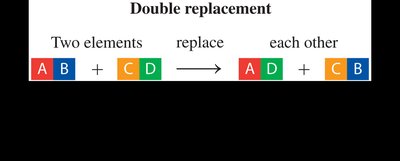
Double Replacement Reaction: The ions of two compounds exchange places to form two new compounds.
Examples of Reaction Types
Combination:
Decomposition:
Single Replacement:
Double Replacement:
Balancing Chemical Equations
Principles of Balancing
A balanced chemical equation has the same number of atoms of each element on both sides of the equation. This reflects the law of conservation of mass, which states that matter is neither created nor destroyed in a chemical reaction.
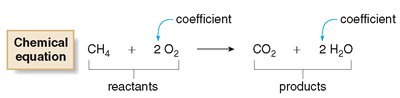
Chemical Equation: Uses chemical formulas and symbols to represent reactants and products.
Coefficients: Whole numbers placed in front of formulas to indicate the number of units involved.
Subscripts: Indicate the number of atoms in a molecule and cannot be changed to balance an equation.

Steps for Balancing Equations
Count the number of atoms of each element on both sides of the equation.
Identify elements that are not balanced.
Balance one element at a time by adjusting coefficients.
Repeat until all elements are balanced.
Check your work to ensure the same number of each atom appears on both sides.
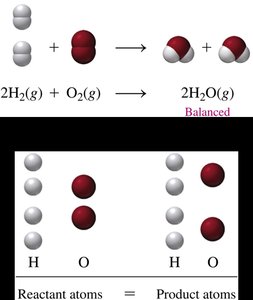
Example: Balancing Water Formation
Unbalanced:
Balance O by making product
Balance H by making reactant
Balanced:
Hints for Balancing Equations
If an equation contains a pure element as a product or reactant, assign that element’s coefficient last.
Save hydrogen and oxygen balancing for last, as they often appear in multiple compounds.
Combustion Reactions
Definition and Characteristics
A combustion reaction involves a carbon-containing compound reacting with oxygen gas to produce carbon dioxide and water, releasing energy as heat.
General Equation: (energy)
Complete Combustion: Produces CO2 and H2O.
Incomplete Combustion: Occurs with insufficient O2, producing CO (carbon monoxide) and H2O.
Example:
Incomplete Combustion Example:
Practice Problems
Balance and classify the following reactions:
Equation | Type | Balanced Form |
|---|---|---|
Fe2O3 + C → Fe + CO | Single Replacement | Fe2O3 + 3C → 2Fe + 3CO |
Al + FeO → Fe + Al2O3 | Single Replacement | 2Al + 3FeO → 3Fe + Al2O3 |
Summary Table: Reaction Types
Type | General Equation | Description |
|---|---|---|
Combination | A + B → AB | Two or more reactants form one product |
Decomposition | AB → A + B | One reactant splits into two or more products |
Single Replacement | A + BC → AC + B | One element replaces another in a compound |
Double Replacement | AB + CD → AD + CB | Ions in two compounds exchange places |
Combustion | Hydrocarbon + O2 → CO2 + H2O | Reaction with O2 producing CO2 and H2O |
Additional info: These notes cover the core concepts from Ch.7 Chemical Quantities and Reactions, including reaction classification, balancing equations, and combustion reactions, as outlined in the course syllabus.
