 Back
BackChemical Reactions and Oxidation-Reduction: A Mini-Textbook Study Guide
Study Guide - Smart Notes

5.3 Overview of Chemical Reactions
Types of Chemical Reactions
Chemical reactions are classified based on how reactants transform into products. Understanding these types is fundamental for predicting reaction outcomes and energy changes.
Synthesis (Combination) Reactions: Two or more reactants combine to form a single product. These reactions often require energy input, as the product is less random (higher free energy) than the reactants. General form: Example:
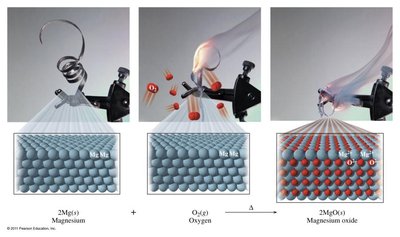
Decomposition Reactions: A single reactant breaks down into two or more products. These reactions often release energy, as stronger bonds form in the products. General form: Example:
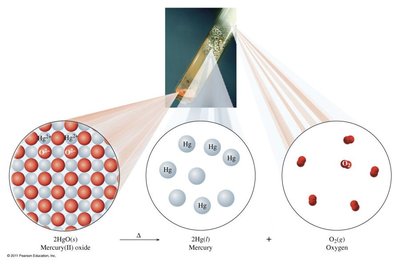
Single Replacement (Displacement) Reactions: One element replaces another in a compound. General form: Example:
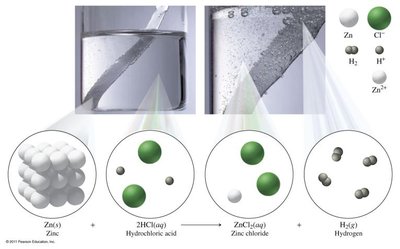
Double Replacement Reactions: Two compounds exchange ions to form two new compounds, often resulting in a gas, solid, or colored complex. General form: Example:
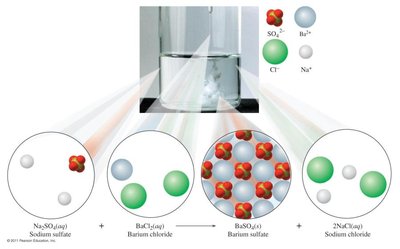
Combustion Reactions: An organic compound reacts with oxygen to produce carbon dioxide and water, releasing heat. General form: Example:

Recognizing Reaction Types
Combination: Two or more reactants form one product.
Decomposition: One reactant forms two or more products.
Single Replacement: One element replaces another in a compound.
Double Replacement: Two compounds exchange ions.
Combustion: Organic compound reacts with oxygen, forms CO2 and H2O.
Reversible and Irreversible Reactions
Irreversible Reactions
Irreversible reactions proceed in only one direction, typically because the products are much lower in energy than the reactants (large negative ΔG).
Example: Combustion reactions, such as burning ethanol.
Reversible Reactions and Chemical Equilibrium
Reversible reactions can proceed in both directions. When the rates of the forward and reverse reactions are equal, the system is at equilibrium (ΔG = 0).
Equilibrium: The concentrations of reactants and products remain constant, but are not necessarily equal.
Organic and Biochemical Reactions
Organic Reactions
Organic reactions focus on changes in functional groups. The connectivity of atoms is shown, and small molecules or reaction conditions are indicated above or below the reaction arrow.
Biochemical Reactions
Biochemical reactions are written similarly to organic reactions. Often, energy transfer is shown at the reaction arrow, indicating coupled reactions.
Oxidation and Reduction (Redox) Reactions
Definition and Importance
Oxidation-reduction reactions involve the transfer of electrons between atoms or molecules. These reactions are essential for energy production, corrosion, and many biological processes.
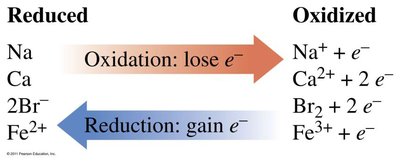
Oxidation: Loss of electrons; atom becomes more positive.
Reduction: Gain of electrons; atom becomes more negative.
Reducing Agent: The species that is oxidized.
Oxidizing Agent: The species that is reduced.

Transfer of Electrons
Elements in their pure form have a neutral charge.
Example: in (lost 3 electrons)
Example: (gained 2 electrons)
Oxidation-Reduction in Various Reaction Types
Occurs in combination, decomposition, single replacement, and combustion reactions.
Oxidation: Loss of electrons (OIL).
Reduction: Gain of electrons (RIG).
Example: Single Replacement Redox Reaction
Consider the reaction:
Zinc is oxidized:
Hydrogen is reduced:

Identifying Redox Reactions
In inorganic chemistry, focus on electron movement.
In organic and biochemistry, focus on addition/removal of oxygen and hydrogen.
Adding bonds to oxygen (or nitrogen) is oxidation; adding bonds to hydrogen is reduction.
Organic Redox Reactions
Oxidation and Reduction in Organic Molecules
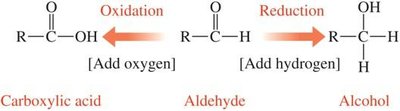
Organic molecules are oxidized if they gain oxygen or lose hydrogen, and reduced if they gain hydrogen or lose oxygen. These changes are often visualized by tracking bonds to oxygen and hydrogen.
Aldehydes: Can be oxidized to carboxylic acids or reduced to alcohols.
Example: Oxidation of aldehyde to carboxylic acid; reduction to alcohol.
Oxidation-Reduction in Metabolism
Role in Cellular Energy Production
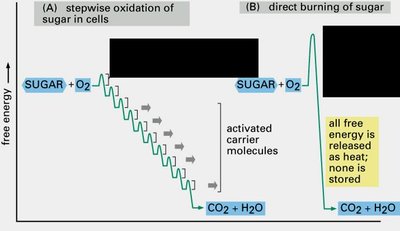
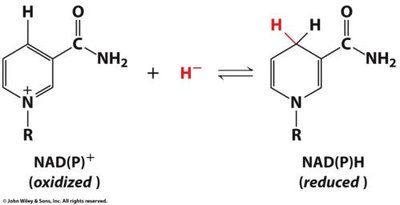
Redox reactions are central to metabolism. As metabolites are oxidized, electrons are transferred to carriers such as NAD+ or NADP+. Subsequent oxidation of these carriers generates ATP, the cell's energy currency.
Example: Glucose oxidation in aerobic respiration.
Electron carriers: NAD+, NADP+, cytochrome c.
Biochemical Redox Reactions
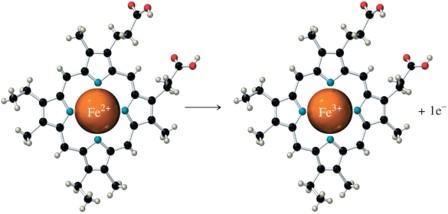
Metals in biological systems, such as iron in cytochrome c, undergo oxidation and reduction. Cytochrome c is crucial for ATP production, with iron cycling between Fe2+ and Fe3+ states.
Summary Table: Types of Chemical Reactions
Type | General Equation | Key Feature | Example |
|---|---|---|---|
Synthesis | Single product | ||
Decomposition | Single reactant | ||
Single Replacement | One element replaces another | ||
Double Replacement | Exchange ions | ||
Combustion | Organic compound + O2 |
