 Back
BackChemical Reactions: Mole and Mass Relationships (Stoichiometry)
Study Guide - Smart Notes

Chapter 6: Chemical Reactions – Mole and Mass Relationships
Introduction to Stoichiometry
Stoichiometry is the study of the quantitative relationships between the amounts of reactants and products in a chemical reaction. It is grounded in the laws of conservation of mass and energy, ensuring that the total mass and number of atoms remain constant throughout a reaction.
The Mole and Avogadro’s Number
Definition and Importance of the Mole
Mole (mol): The amount of a substance that contains as many particles (atoms, molecules, ions) as there are atoms in exactly 12 grams of carbon-12.
Avogadro’s Number (NA): particles per mole.
The mole concept allows chemists to count and compare quantities of extremely small particles using measurable masses.
It is essential for balancing chemical equations and ensuring the law of conservation of mass is satisfied.

Analogy and Application
The term "mole" is analogous to terms like "dozen" (12 items) or "pair" (2 items), but represents items.
One mole of any element contains atoms of that element.

Molar Mass and Molecular Weight
Definitions
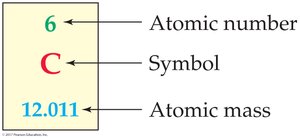
Atomic Mass: The average mass of an element’s atoms, based on the natural abundance of isotopes.
Molecular Weight (MW): The sum of the atomic masses of all atoms in a molecule.
Molar Mass: The mass in grams of one mole of a substance, numerically equal to its molecular or formula weight (g/mol).
Calculating Molar Mass
Find the atomic mass of each element in the compound (from the periodic table).
Multiply by the number of atoms of each element in the formula.
Add the total masses to obtain the molar mass.
Example: For Al2(SO4)3:
2 Al × 26.98 g/mol + 3 S × 32.07 g/mol + 12 O × 16.00 g/mol = 342.17 g/mol
Using the Mole in Chemical Calculations
Conversions Involving Moles
To convert between grams and moles, use the molar mass as a conversion factor.
To convert between moles and number of particles, use Avogadro’s number.
Key Equations:
Grams to moles:
Moles to particles:
Note: You cannot convert directly from grams to particles; you must first convert grams to moles.
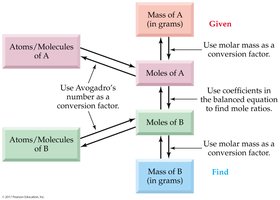
Mole Map
The mole map visually summarizes the relationships between mass, moles, and number of particles, and how to use conversion factors in calculations.
Mole Relationships in Chemical Compounds
Subscripts and Moles
The subscripts in a chemical formula indicate the number of atoms of each element in one molecule and the number of moles of each element in one mole of the compound.
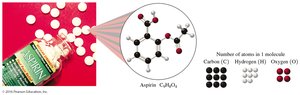
Example: Aspirin (C9H8O4):
1 molecule: 9 C, 8 H, 4 O atoms
1 mole: 9 mol C, 8 mol H, 4 mol O
Stoichiometry in Chemical Equations
Coefficients and Mole Ratios
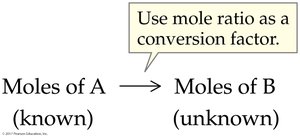
Coefficients in balanced chemical equations represent the number of moles of each reactant and product.
Mole ratios derived from coefficients are used as conversion factors in stoichiometric calculations.

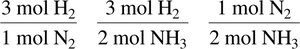
Types of Stoichiometric Calculations
Mole-to-mole: Use mole ratios from the balanced equation.
Mole-to-mass and mass-to-mole: Use molar mass as a conversion factor.
Mass-to-mass: Convert mass to moles, use mole ratio, then convert back to mass.

Limiting Reagent and Percent Yield
Limiting Reagent
The limiting reagent is the reactant that is completely consumed first, thus limiting the amount of product formed.
To identify the limiting reagent, compare the mole ratios of reactants used to those required by the balanced equation.
Theoretical, Actual, and Percent Yield
Theoretical yield: The maximum amount of product that can be formed from the limiting reagent.
Actual yield: The amount of product actually obtained from a reaction (often less than theoretical due to losses).
Percent yield: The ratio of actual yield to theoretical yield, expressed as a percentage.
Percent Yield Formula:

Practice and Application
Sample Problems
Calculate the molar mass of a compound given its formula.
Convert between grams, moles, and number of particles for a given substance.
Use balanced chemical equations to determine the amounts of reactants and products.
Identify the limiting reagent in a reaction and calculate theoretical and percent yields.
Summary Table: Key Stoichiometric Relationships
Type of Conversion | Conversion Factor | Example |
|---|---|---|
Grams ↔ Moles | Molar Mass (g/mol) | |
Moles ↔ Particles | Avogadro’s Number | |
Moles A ↔ Moles B | Mole Ratio (from equation) |
Additional info: Laboratory exercises such as synthesis and decomposition of copper(II) oxide, and limiting reactant experiments, reinforce these concepts through hands-on practice.
