 Back
BackChemistry and Measurement: Classification of Matter, Unit Conversion, and Density
Study Guide - Smart Notes

Chemistry and Measurement
Units of Measurement
Understanding units of measurement is fundamental in chemistry, as it allows for accurate quantification and comparison of substances. The International System of Units (SI) is commonly used, with mass measured in kilograms (kg) and grams (g), and temperature in Kelvin (K) and degrees Celsius (°C).
Mass Conversion: 1 kg = 1000 g. To convert kilograms to grams, multiply by 1000.
Example: A 2.5 kg sample is equivalent to 2,500 g.
Temperature Conversion: The SI unit for temperature is Kelvin (K). Conversion formulas:
Example: 15°C = 288.15 K; 314 K = 40.85°C
Significant Figures and Calculations
Significant figures reflect the precision of a measurement. When performing calculations, the number of significant figures in the result should match the least precise measurement used in the calculation.
Rules: Nonzero digits are always significant; zeros between nonzero digits are significant; leading zeros are not significant; trailing zeros in a decimal number are significant.
Prefixes and Equalities
Prefixes are used to express multiples or fractions of units. Common prefixes include kilo- (k, 103), centi- (c, 10-2), and milli- (m, 10-3).
Example: 1 g = 1000 mg
Writing Conversion Factors
Conversion factors are ratios used to convert from one unit to another. Place the desired unit in the numerator and the given unit in the denominator.
Example: To convert 126 mg to g:
Matter and Energy
Classification of Matter
Matter is anything that has mass and occupies space. It can be classified by its state (solid, liquid, gas) and its composition (pure substances or mixtures).
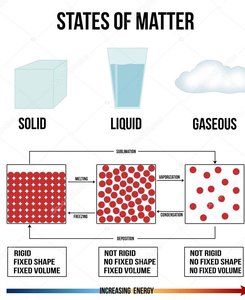
States of Matter:
Solid: Definite shape and volume; particles are very close together.
Liquid: Definite volume, indefinite shape; particles are close together but can move.
Gas: Indefinite shape and volume; particles are far apart and move freely.
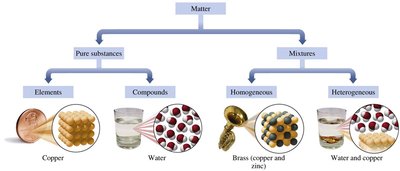
Composition:
Pure Substance: Fixed composition; can be an element (e.g., O2, Cu) or a compound (e.g., H2O, NaCl).
Mixture: Combination of two or more pure substances; can be homogeneous (uniform throughout, e.g., air) or heterogeneous (composition varies, e.g., oil and water).
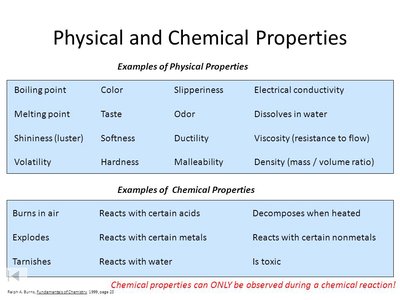
Physical and Chemical Properties
Physical properties are characteristics observed or measured without changing the identity of a substance, such as shape, state, boiling and freezing points, density, and color. Physical changes occur when matter undergoes a change of state, but its composition remains constant. Chemical properties result in a change in composition and can be observed only through a chemical reaction.
Examples of Physical Properties: Boiling point, melting point, density, color.
Examples of Chemical Properties: Flammability, decomposition, toxicity.
Changes of State
Matter undergoes changes of state when it is converted from one state to another, such as melting, freezing, vaporization, condensation, sublimation, and deposition. Each change of state occurs at a specific temperature for a given substance.
Melting Point: Temperature at which a solid turns to liquid.
Boiling Point: Temperature at which a liquid turns to gas.
Freezing Point: Temperature at which a liquid turns to solid.

Density
Definition and Calculation
Density is the ratio of mass to volume and is used to characterize substances, as each has a unique density. The formula for density is:
Units: g/mL for liquids, g/cm3 for solids
Rearranged formulas:

Applications and Examples
Density is used to determine whether an object will float or sink in a medium, and to characterize substances in laboratory and clinical settings.
Example: A blood sample with a mass of 5.30 g and a volume of 5.00 mL has a density of .
Example: The volume of a liquid with a density of 5.20 g/mL and a mass of 1.55 g is .
Example: The mass of a red blood cell with a density of 1.125 g/mL and a volume of mL is g.
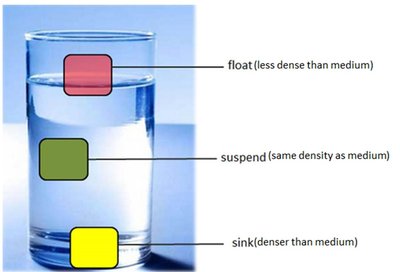
Density in Practice
Density can be used to analyze samples in medical and scientific contexts, such as determining bone density in osteoporosis or the density of blood samples.
Example: A bone sample with a mass of 2.15 g and a volume of 1.40 cm3 has a density of .

Problem Solving and Practice
Unit Conversion Practice
Converting between units is a key skill in chemistry. Always use the appropriate conversion factor and ensure the desired unit is in the numerator.
Example: 3 tablets contain 126 mg of propranolol. To convert to grams:
Density Practice
Calculating density, mass, or volume requires careful use of the formulas and attention to units and significant figures.
Example: If a liquid has a density of 5.20 g/mL and a mass of 1.55 g, its volume is .
Summary Table: States of Matter
Characteristic | Solid | Liquid | Gas |
|---|---|---|---|
Shape | Definite | Indefinite | Indefinite |
Volume | Definite | Definite | Indefinite |
Arrangement of Particles | Fixed, very close | Random, close | Random, far apart |
Interaction between Particles | Very strong | Strong | Essentially none |
Movement of Particles | Very slow | Moderate | Very fast |
Examples | Ice, salt, iron | Water, oil, vinegar | Water vapor, helium, air |

