 Back
BackChemistry and Measurements: Essential Concepts and Applications
Study Guide - Smart Notes

Ch.2 Chemistry and Measurements
SI Units and Base Quantities
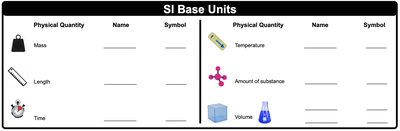
The International System of Units (SI) is the standard system used in science for measurement. It is based on seven base units, but chemistry commonly uses six: mass, length, time, temperature, amount of substance, and volume. Each physical quantity has a specific unit and symbol.
Mass: Measured in kilograms (kg)
Length: Measured in meters (m)
Time: Measured in seconds (s)
Temperature: Measured in kelvin (K)
Amount of substance: Measured in moles (mol)
Volume: Measured in cubic meters (m3), but liters (L) are commonly used in chemistry




Example: The SI unit for volume is the cubic meter (m3), but in laboratory settings, liters (L) and milliliters (mL) are more common.
Significant Figures
Significant figures (sig figs) are the digits in a measurement that are known with certainty plus one estimated digit. They reflect the precision of a measurement.
Exact Numbers: Values obtained by counting or defined quantities (e.g., 12 eggs in a dozen) have an infinite number of significant figures.
Inexact Numbers: Values obtained by measurement, which include some uncertainty (e.g., 12.53 inches).
Rules for Significant Figures:
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Leading zeros are not significant.
Trailing zeros in a number with a decimal point are significant.
Trailing zeros in a whole number without a decimal point are not significant.
For numbers in scientific notation, all digits in the coefficient are significant.
Example: 0.003840 has four significant figures.
Precision in Measurements
The number of significant figures in a measurement indicates its precision. When recording measurements, include all known digits plus one estimated digit.
Example: A measurement of 25.00 mL is more precise than 25 mL.


Significant Figures in Calculations
When performing calculations, the number of significant figures in the result depends on the operation:
Multiplication/Division: The result should have the same number of significant figures as the value with the fewest significant figures.
Addition/Subtraction: The result should have the same number of decimal places as the value with the fewest decimal places.
Mixed Operations: Follow the order of operations (PEMDAS: Parentheses, Exponents, Multiplication/Division, Addition/Subtraction).
Example Calculation:
402.09 − 212.2 + 2.671
Metric Prefixes
Metric prefixes are used to express multiples or fractions of base units. They are based on powers of ten and make it easier to express very large or very small quantities.
Kilo- (k):
Centi- (c):
Milli- (m):
Micro- (\mu):
Nano- (n):
Pico- (p):
Example: 694 kg to µg involves converting kilograms to grams, then grams to micrograms.
Conversion Factors and Dimensional Analysis
Conversion factors are ratios that relate two different units. Dimensional analysis is a systematic approach to problem-solving that uses conversion factors to move from one unit to another.
Example: To convert 3 hours to seconds, use the conversion factors 1 hour = 60 minutes and 1 minute = 60 seconds.
General Format:
Given Amount × Conversion Factor(s) = End Amount
Parts Per Million (ppm) and Parts Per Billion (ppb)
These units are used to express very dilute concentrations of substances.
ppm (parts per million):
ppb (parts per billion):
For aqueous solutions: 1 ppm = 1 mg/L, 1 ppb = 1 µg/L
Density
Density is the amount of mass per unit volume. It is a key property for identifying substances and is used in various calculations.
Formula:
Units: g/cm3 or g/mL for solids and liquids; g/L for gases
Density of Geometric Objects
For objects with regular shapes, volume can be calculated using geometric formulas:
Cube:
Sphere:
Cylinder:
Density of Non-Geometric Objects: Water Displacement
For irregularly shaped objects, volume is determined by the amount of water displaced when the object is submerged.
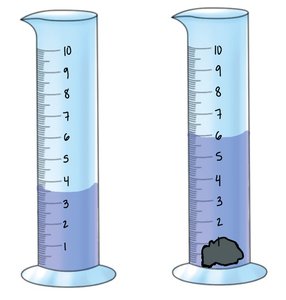
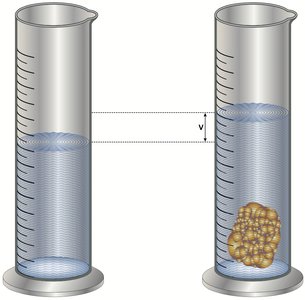
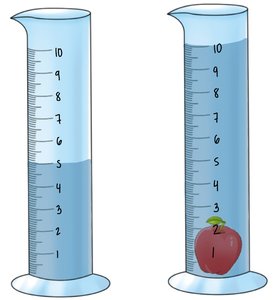
Example: If a solid increases the water level from 200 mL to 260 mL, the volume of the solid is 60 mL.
Specific Gravity
Specific gravity is the ratio of the density of a substance to the density of water at the same temperature. It is a unitless quantity.
Formula:
Example: If the density of sulfuric acid is 1.27 g/mL at 25ºC, its specific gravity is 1.27 (since the density of water at 25ºC is approximately 1.00 g/mL).
Additional info: These concepts are foundational for all subsequent topics in general, organic, and biological chemistry, as accurate measurement and unit conversion are essential for laboratory and theoretical work.
