 Back
BackChemistry Basics for Nursing: Matter, Measurement, and Calculations
Study Guide - Smart Notes

Chemistry Basics: Matter and Measurement
Classification of Matter
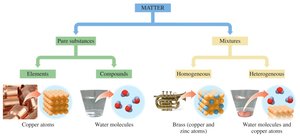
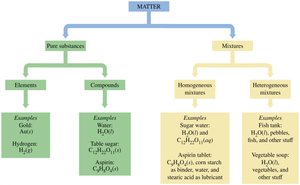
Matter is anything that occupies space and has mass. It can be classified into pure substances and mixtures, each with distinct properties and examples.
Pure Substances: These have a fixed composition and distinct properties. They are further divided into:
Elements: Substances made of only one type of atom (e.g., copper, gold, hydrogen).
Compounds: Substances composed of two or more elements chemically combined in fixed ratios (e.g., water, table sugar).
Mixtures: Physical combinations of two or more substances. They can be:
Homogeneous Mixtures (Solutions): Uniform composition throughout (e.g., sugar water, brass).
Heterogeneous Mixtures: Non-uniform composition (e.g., vegetable soup, mixtures of water and copper atoms).
The Periodic Table and Elements
The periodic table organizes all known elements by increasing atomic number and similar chemical properties. Elements are classified as metals, non-metals, and metalloids. Groups (columns) and periods (rows) help identify element families and trends.
Metals: Good conductors of heat and electricity, malleable, ductile (e.g., iron, copper).
Non-metals: Poor conductors, often gases or brittle solids (e.g., oxygen, nitrogen).
Metalloids: Properties intermediate between metals and non-metals (e.g., silicon).

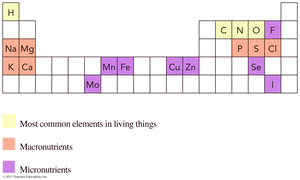
Elements in Living Things
Some elements are especially important for life. The most common elements in living things include carbon, hydrogen, oxygen, and nitrogen. Macronutrients and micronutrients are also essential for biological functions.
Macronutrients: Needed in amounts >100 mg/day (e.g., Na, K, Ca, Mg).
Micronutrients: Needed in smaller amounts (e.g., Fe, Zn, Se, I).
Measurement in Chemistry
Units and SI System
Chemistry uses the International System of Units (SI) for consistency. The main base units are:
Length: meter (m)
Mass: gram (g)
Time: second (s)
Volume: liter (L)
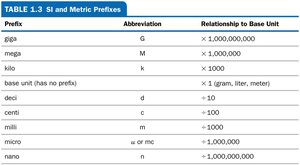
Prefixes are used to indicate multiples or fractions of base units.
Volume and Its Measurement
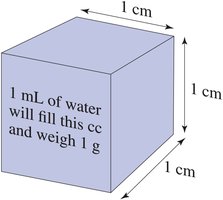
Volume is the amount of space occupied by a substance. Common units include liters (L), milliliters (mL), and cubic centimeters (cm3 or cc). In the lab, volume is measured with graduated cylinders, pipets, or syringes.
1 mL = 1 cm3 = 1 cc
1 tsp ≈ 5 mL

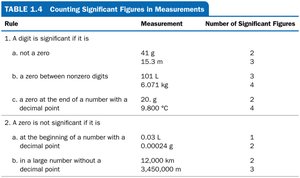
Significant Figures
Significant figures (sig figs) reflect the precision of a measurement. The rules for counting significant figures are:
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Leading zeros are not significant.
Trailing zeros are significant only if there is a decimal point.

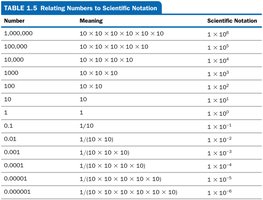
Scientific Notation
Scientific notation expresses very large or small numbers as a coefficient (between 1 and 10) multiplied by a power of ten. Only significant figures are shown in the coefficient.
General form:
Example: 5,370,000 =
Physical and Chemical Changes
Physical Changes
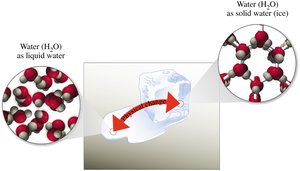
Physical changes alter the form or appearance of matter but do not change its composition. Examples include changes of state (solid, liquid, gas).
Examples: Melting ice, boiling water
Chemical Changes
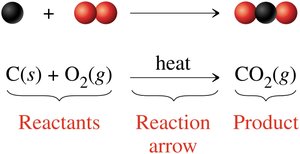
Chemical changes (reactions) result in the formation of new substances with different properties. Bonds between atoms are broken and formed.
Example: Combustion of charcoal (carbon reacts with oxygen to form carbon dioxide)

States of Matter
Solids, Liquids, and Gases
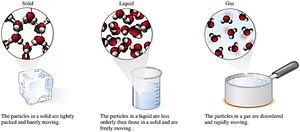
Matter exists in three main states: solid, liquid, and gas. Each state has distinct properties related to shape, volume, and particle arrangement.
Solids: Definite shape and volume; particles are tightly packed and vibrate in place.
Liquids: Definite volume, take the shape of their container; particles are less orderly and move freely.
Gases: No definite shape or volume; particles are far apart and move rapidly.
Dimensional Analysis and Unit Conversions
Dimensional analysis uses conversion factors to change units. Units are treated like numbers and can be multiplied, divided, or canceled.
Example: To convert 20 minutes to hours, use the conversion factor .
Accuracy and Precision
Accuracy refers to how close a measurement is to the true value. Precision refers to how closely repeated measurements agree with each other. More significant figures indicate greater precision.
Percentages in Chemistry and Health
Percentages express quantities as parts per hundred. They are used in health for medication dosages, nutrition labeling, and more.

Temperature Scales
Temperature is measured in Fahrenheit (°F), Celsius (°C), or Kelvin (K). The Kelvin scale is the SI unit for temperature.
Normal body temperature: 37°C (98.6°F)
Hyperthermia: >40°C (104°F)
Hypothermia: <35°C (95°F)


Energy and Heat
Energy is the capacity to do work or produce heat. Heat is a form of energy transfer due to temperature difference. Specific heat is the amount of heat required to raise the temperature of 1 g of a substance by 1°C.
Water has a high specific heat (1.00 cal/g·°C), which helps regulate body temperature.

Units and Dosing in Health Care
Health professionals use both SI (metric) and U.S. customary units. Accurate unit conversions are essential for safe medication dosing.
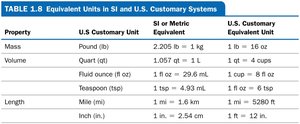
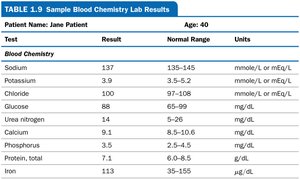
Reading Lab Reports
Lab reports provide important clinical data, such as blood chemistry values. Understanding units and normal ranges is crucial for interpreting results.
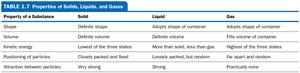
Summary Table: Properties of Solids, Liquids, and Gases
Property | Solid | Liquid | Gas |
|---|---|---|---|
Shape | Definite | Adopts shape of container | Adopts shape of container |
Volume | Definite | Definite | Fills volume of container |
Kinetic energy | Lowest | More than solid, less than gas | Highest |
Particle arrangement | Closely packed, fixed | Loosely packed, random | Far apart, random |
Attractive forces | Very strong | Strong | Practically none |
