 Back
BackChemistry Basics: Matter and Measurement (Chapter 1 Study Guide)
Study Guide - Smart Notes

Chemistry Basics—Matter and Measurement
What is Matter?
Chemistry is the study of matter, which is anything that occupies space and has mass. Understanding the classification and properties of matter is fundamental to all chemical sciences.
Matter: Anything that takes up space and has mass.
Examples: Water, air, rocks, living organisms.

Classifying Matter: Pure Substance or Mixture
Types of Matter
Matter can be classified as either pure substances or mixtures. Pure substances have a uniform composition, while mixtures contain two or more substances physically combined.
Pure Substance: Made up of only one type of substance; represented by a single chemical formula or symbol.
Mixture: Combination of two or more substances; can be separated into individual components.
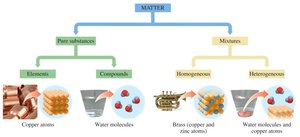
Pure Substances: Elements and Compounds
Pure substances are further divided into elements and compounds. Elements consist of only one type of atom, while compounds are made of two or more elements chemically bonded in fixed ratios.
Element: Simplest form of matter; only one type of atom.
Atom: Smallest unit of an element retaining its properties.
Compound: Pure substance formed from two or more elements chemically joined.
Chemical Formula: Shows the types and numbers of atoms in a compound (e.g., for water).
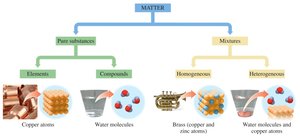
Mixtures: Homogeneous and Heterogeneous
Mixtures can be homogeneous (uniform composition) or heterogeneous (non-uniform composition). Homogeneous mixtures are also called solutions.
Homogeneous Mixture: Uniform throughout; e.g., saltwater.
Heterogeneous Mixture: Not uniform; e.g., salad.
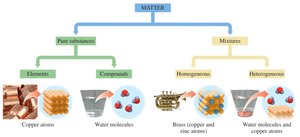
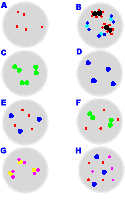
Visual Classification of Matter
Visual representations help identify pure elements, pure compounds, and various mixtures. Each type can be distinguished by the arrangement and types of particles present.
Pure elements: Only one type of atom.
Pure compounds: Only one type of molecule, made from two or more elements.
Mixtures: Contain more than one type of particle (atoms or molecules).
Elements, Compounds, and the Periodic Table
The Periodic Table
The periodic table organizes elements by increasing atomic number and groups elements with similar properties into columns called groups or families. Rows are called periods.
Period: Horizontal row.
Group: Vertical column.
Atomic Number: Number of protons in an atom.
Symbol: One- or two-letter abbreviation for an element.
Atomic Weight: Average mass of an atom in atomic mass units (amu).



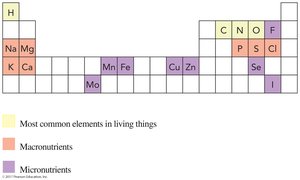
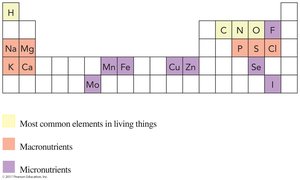
Elements Essential for Human Health
Certain elements are required for human health, classified as macronutrients and micronutrients based on the amount needed daily.
Macronutrients: Needed in amounts > 100 mg/day (e.g., Na, K, Ca).
Micronutrients: Needed in amounts < 100 mg/day (e.g., Fe, Zn, Cu).
Most common elements in living things: H, C, N, O, P, S.
How Matter Changes: Physical and Chemical Changes
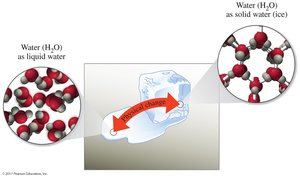
Physical Change
A physical change alters the form or appearance of matter without changing its chemical identity. Common examples include changes of state (solid, liquid, gas).
Physical Change: No new substance is formed; identity remains the same.
Examples: Melting, freezing, dissolving.
Chemical Change
A chemical change results in the formation of new substances with different chemical properties. This process is called a chemical reaction.
Chemical Change: Chemical identity changes; new substances are formed.
Example: Burning charcoal produces new compounds.

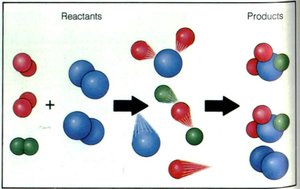
Chemical Reactions
During a chemical reaction, bonds in reactants are broken and new bonds are formed to create products. Atoms are rearranged, but the total mass remains constant.
Reactants: Starting substances in a reaction.
Products: New substances formed.
Law of Conservation of Mass: Matter is not created or destroyed in a chemical reaction.
Chemical Equation: Represents the reactants and products, including their physical states (s, l, g, aq).
Example of a balanced chemical equation:
Balancing Chemical Equations: Add coefficients to ensure the number of atoms of each element is the same on both sides of the equation.
Summary Table: Classification of Matter
Type | Description | Example |
|---|---|---|
Element | Only one type of atom | Oxygen (O), Copper (Cu) |
Compound | Two or more elements chemically joined | Water (H2O), Carbon dioxide (CO2) |
Homogeneous Mixture | Uniform composition | Saltwater, air |
Heterogeneous Mixture | Non-uniform composition | Salad, granite |
