 Back
BackChemistry Basics: Matter, Measurement, and Atomic Structure
Study Guide - Smart Notes

Chemistry Basics - Matter and Measurement
Introduction to Chemistry
Chemistry is the scientific study of matter, its properties, and the changes it undergoes. Matter is anything that occupies space and has mass.
Matter: Takes up space (volume) and has weight (mass).
Chemistry: Explores the composition, structure, and properties of substances.


Classifying Matter
Matter can be classified into pure substances and mixtures. Pure substances include elements and compounds, while mixtures can be homogeneous or heterogeneous.
Pure Substance: Made up of only one type of substance.
Element: Contains only one type of atom.
Compound: Contains two or more elements joined together in a fixed ratio.
Mixture: Combination of two or more pure substances.
Homogeneous Mixture: Same composition throughout.
Heterogeneous Mixture: Composition varies.
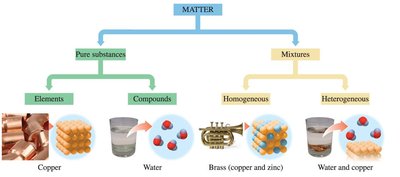
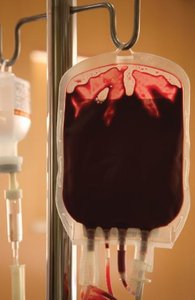
Examples of Matter Classification
Blood: Example of a mixture.
Diamond: Example of a pure substance (element).
Sugar: Example of a pure substance (compound).


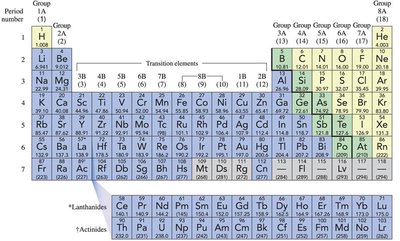
Periodic Table of Elements
The periodic table organizes elements based on their atomic number and properties. Each element is represented by a unique symbol.
Element Symbols: Consist of one or two letters.
Groups: Vertical columns, elements in the same group have similar properties.
Periods: Horizontal rows, elements in the same period have the same number of electron shells.

Atomic Structure
Structure of the Atom
Atoms are the smallest units of matter that retain the chemical properties of an element. They are composed of three basic subatomic particles:
Proton: Positive charge (+1), mass = 1 amu, located in the nucleus.
Neutron: No charge (0), mass = 1 amu, located in the nucleus.
Electron: Negative charge (-1), negligible mass, located outside the nucleus.
Particle | Charge | Mass (amu) | Location |
|---|---|---|---|
Proton | +1 | 1 | Nucleus |
Neutron | 0 | 1 | Nucleus |
Electron | -1 | negligible | Outside nucleus |
*Additional info: Atoms are electrically neutral because the number of protons equals the number of electrons.*
Atomic Number and Isotopes
Atomic Number: Number of protons in the nucleus; identifies the element.
Isotopes: Atoms of the same element with different numbers of neutrons.
Mass Number: Sum of protons and neutrons in an atom.
Atomic Mass: Average mass of an element, considering the relative abundance of its isotopes.
Measurement and Scientific Notation
Scientific Notation
Scientific notation is a shorthand method for writing very large or very small numbers. The general form is:
C is a number between 1 and 9 (shows significant figures).
i is the exponent indicating the number of tens places.
Significant Figures
Significant figures reflect the precision of a measurement. Rules for counting significant figures:
Nonzero digits are always significant.
Zeros are significant only if they are between nonzero digits or at the end of a number with a visible decimal point.
Metric System and Unit Conversions
The metric system uses base units and prefixes to express measurements. Common base units include grams (g), liters (L), and meters (m).
Prefix | Symbol | Relationship to Base Unit |
|---|---|---|
giga | G | 1,000,000,000 (10^9) |
mega | M | 1,000,000 (10^6) |
kilo | k | 1,000 (10^3) |
deci | d | 0.1 (10^-1) |
centi | c | 0.01 (10^-2) |
milli | m | 0.001 (10^-3) |
micro | μ | 0.000001 (10^-6) |
nano | n | 0.000000001 (10^-9) |
*Additional info: Conversion factors are used to convert between units, e.g., 1 in. = 2.54 cm.*
The Mole and Molar Mass
The Mole Concept
A mole is a unit for counting particles. One mole contains particles (Avogadro's number).
1 mole of H atoms = H atoms
1 mole of O atoms = O atoms
Molar Mass
Molar mass is the mass (in grams) of one mole of a substance. It is calculated using atomic masses from the periodic table.
Example: Molar mass of H2O = 2 × 1.0079 (H) + 1 × 15.9994 (O) = 18.0152 g/mol
Electron Arrangement and Bonding
Electron Shells and Valence Electrons
Electrons are arranged in shells (energy levels) around the nucleus. The outermost electrons are called valence electrons and are involved in bonding.
Shell 1: max 2 electrons
Shell 2: max 8 electrons
Shell 3: max 18 electrons
Shell 4: max 32 electrons
Lewis Dot Diagrams and the Octet Rule
Lewis dot diagrams represent valence electrons as dots around the element symbol. Most atoms strive to achieve 8 valence electrons (octet rule), except for H (2), B (6), and He (2).
Atoms achieve octet by sharing electrons (covalent bonds).
Covalent Bonds and Molecules
A covalent bond is formed when two atoms share a pair of electrons. Covalent compounds are generally formed when nonmetals share electrons, resulting in molecules with fixed composition.
Example: Water (H2O) is a covalent compound.
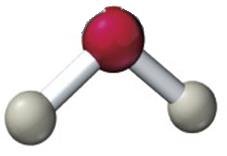
Molecular Geometry and Polarity
VSEPR Theory and Molecular Shape
The shape of a molecule is determined by the repulsion of valence electron pairs (VSEPR theory). Common shapes include linear, bent, trigonal planar, tetrahedral, and trigonal pyramidal.
Example: Water (H2O) has a bent shape due to two lone pairs on oxygen.
Bond Polarity and Molecule Polarity
Nonpolar Covalent Bond: Equal sharing of electrons.
Polar Covalent Bond: Unequal sharing of electrons, creating dipoles.
Electronegativity: Atom's attraction for shared electrons in a bond.
Molecule polarity depends on both bond polarity and molecular geometry.
States of Matter and Intermolecular Forces
Intermolecular Forces
Intermolecular forces are attractions between molecules, weaker than covalent bonds. Types include:
London Forces: Temporary induced dipoles, present in all molecules.
Dipole-Dipole Attractions: Permanent dipoles, present in polar molecules.
Hydrogen Bonding: Strong dipole-dipole force, requires H bonded to N, O, or F.
States of Matter
Solid: Fixed shape and volume.
Liquid: Fixed volume, no fixed shape.
Gas: No fixed shape or volume, highly compressible.
Changes of State
Phase changes (melting, boiling) occur when enough energy is supplied to overcome intermolecular forces.
Covalent bonds are not broken during phase changes.
Behavior of Gases and Boyle's Law
Gas Properties
Gases exert pressure due to collisions between molecules and their surroundings.
Pressure units: 1 atm = 760 mm Hg = 760 torr = 29.92 in. Hg = 101,325 pascals.
Boyle's Law
Boyle's Law states that at constant temperature and fixed moles, pressure and volume are inversely proportional:
Increasing pressure decreases volume, and vice versa.
Summary Table: Classification of Matter
Type | Definition | Example |
|---|---|---|
Element | One type of atom | Copper, Diamond |
Compound | Two or more elements in fixed ratio | Water, Sugar |
Homogeneous Mixture | Uniform composition | Brass (copper and zinc) |
Heterogeneous Mixture | Variable composition | Blood, Water and copper |

