 Back
BackChemistry Fundamentals: Matter, Measurements, Atomic Structure, and Nuclear Chemistry
Study Guide - Smart Notes

Chemistry in Our Lives
What is Chemistry?
Chemistry is the study of matter, its transformations, and the energy changes that accompany those transformations. It helps explain everyday phenomena such as color, taste, smell, and the distinction between poisons and medicines. Matter is anything that occupies space and has mass, and it is composed of atoms or groups of atoms called compounds.
Energy: The ability to do work or accomplish change.
Mass: Measurement of the amount of matter in an object.
Atoms: The basic units of matter.
Compounds: Groups of atoms bonded together.
Example: Carbon monoxide (CO) and carbon dioxide (CO2) are both colorless, odorless gases, but differ in their atomic composition and properties.
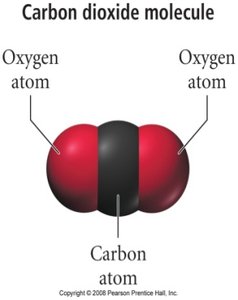
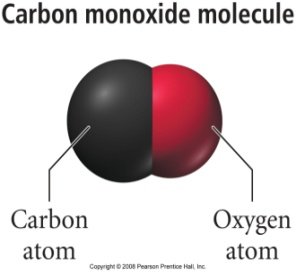
The Scientific Method
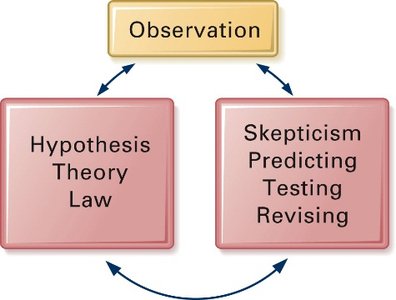
The scientific method is a systematic process used by scientists to make observations, gather data, and explain natural phenomena. It involves forming hypotheses, testing predictions, and revising theories based on evidence.
Observation: Gathering information about the world.
Hypothesis: A tentative explanation for an observation.
Theory: A well-substantiated explanation.
Law: A statement describing consistent natural phenomena.
Chemistry and Measurements
Experimentation & Measurements
Experimentation distinguishes science from pseudoscience. Accurate measurements are essential for collecting reliable data. Every measurement must have a number (with correct significant figures) and a unit.
Accuracy: How close a measurement is to the true value.
Precision: How well repeated measurements agree with each other.
Uncertainty: All measurements have some degree of uncertainty.
Significant Figures: Digits known accurately plus one estimated digit.
Significant Figures in Measurements
Significant figures reflect the precision of a measurement. The number of significant figures is determined by the measuring device and the method used.
Record all accurate numbers plus one estimated number.
Estimate one decimal place past the smallest mark on the measuring device.

Exact Numbers
Exact numbers are whole numbers with an infinite number of significant figures. They apply to counted items or defined quantities (e.g., 12 in = 1 ft).
Rules for Identifying Significant Figures
Start numbering from the left with the first non-zero digit.
Zeros between non-zero digits are significant.
Zeros to the right of a decimal point are significant.
Zeros used as placeholders are not significant unless specified.
Rounding Numbers
If the first digit to be dropped is less than 5, do not round up.
If the first digit to be dropped is 5 or greater, round up.
Significant Figures in Calculations
Addition/Subtraction: The answer should have no more decimal places than the least precise number.
Multiplication/Division: The answer should have the same number of significant figures as the measurement with the least number of significant figures.
Scientific Notation
Scientific notation is used to express very large or very small numbers in the form .
Only one digit to the left of the decimal point.
Positive exponent: large number (decimal moved left).
Negative exponent: small number (decimal moved right).
Matter and Energy
Measurements of Mass and Volume
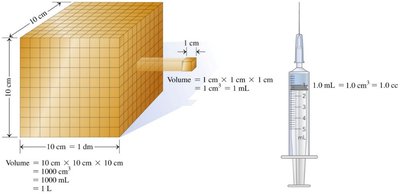
Mass is the amount of matter present in an object. Volume is the amount of space occupied by an object, often measured in cubic centimeters (cm3), milliliters (mL), or cubic centimeters (cc).
1 cm3 = 1 mL = 1 cc
Solids: g/cm3
Liquids: g/mL
Gases: g/L
Temperature
Temperature measures the degree of hotness and the kinetic energy of a substance. Common units are Celsius (°C), Fahrenheit (°F), and Kelvin (K).
Density
Density is the ratio of mass to volume, indicating how much matter is contained in a given volume.
Units: g/cm3 (solids), g/mL (liquids), g/L (gases)

Chemistry and Measurements: The Metric System
Metric System and Prefixes
The metric system is based on factors of ten and uses base units for length (meter), mass (gram), volume (liter), and time (second). Prefixes indicate multiples or fractions of base units.
Kilo- (k):
Centi- (c):
Milli- (m):
Micro- (µ):
Nano- (n):
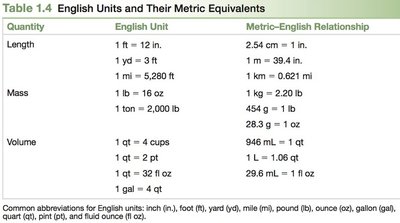
English and Metric Conversion Factors
Conversion factors are used to change one unit to another. English to English conversions are exact, while English to metric conversions are not exact and require attention to significant figures.
Quantity | English Unit | Metric-English Relationship |
|---|---|---|
Length | 1 ft = 12 in. | 2.54 cm = 1 in. |
Mass | 1 lb = 16 oz | 1 kg = 2.20 lb |
Volume | 1 qt = 4 cups | 946 mL = 1 qt |
Matter and Its Classification
Types of Matter
Matter is anything that occupies space and has mass. It is composed of atoms, which may bond to form molecules or large arrays of ions.
Atoms: Smallest units with the characteristics of an element.
Compounds: Smallest units of two or more atoms bonded together.
Ions: Positively or negatively charged atoms or groups of atoms.
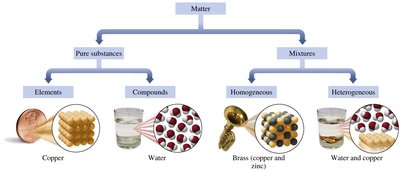
Classification of Matter
Matter can be classified as pure substances or mixtures.
Pure Substances: Constant composition and fixed properties.
Elements: Cannot be broken down by simple chemical means.
Compounds: Can be broken down into two or more elements.
Mixtures: Physical mixtures of two or more substances.
Homogeneous Mixtures: Uniform in appearance and properties.
Heterogeneous Mixtures: Two or more physically distinct phases.
States and Properties of Matter
Physical States of Matter
Matter exists in three primary states: solid, liquid, and gas. Each state has unique properties regarding shape, volume, and particle distance.
Solid: Definite shape and volume; particles are close together.
Liquid: Definite volume, shape defined by container; particles are moderately spaced.
Gas: Shape and volume defined by container; particles are far apart.
Physical and Chemical Properties
Physical properties can be observed or measured without changing the chemical composition. Chemical properties indicate the ability of a substance to convert to new substances during a chemical reaction.
Physical Properties: Color, state, odor, volume, density, melting/boiling/freezing point, viscosity.
Chemical Properties: Ability to undergo chemical reactions (e.g., burning, fermenting).
Physical and Chemical Changes
Physical Change: Alters physical properties without changing chemical composition (e.g., melting, dissolving).
Chemical Change: Converts one substance into another through reorganization of atoms (e.g., burning paper).
Energy and Thermodynamics
Law of Conservation of Energy
Energy cannot be created or destroyed; it can only be transferred. The main forms of energy in chemistry are heat, work, and energy.
Joule (J): SI unit of energy.
Calorie (cal): 1 cal = 4.184 J.
Nutrition Calorie (Cal): 1 Cal = 1000 cal = 1 kcal.
Energy and Nutrition
Food provides energy for the body. Carbohydrates, fats, and proteins release different amounts of energy when metabolized.
Type of Food Molecule | Dietary Calorie Content (kcal/gram) |
|---|---|
Carbohydrate | 4 |
Fat | 9 |
Protein | 4 |
Specific Heat and Calorimetry
Specific heat is the amount of heat energy required to raise the temperature of one gram of a substance by 1°C. Calorimetry measures heat energy changes in chemical reactions.
Where is heat energy, is mass, is specific heat, and is change in temperature.
Atoms and Elements
The Periodic Table
The periodic table organizes elements by increasing atomic number. Elements are grouped into periods (rows) and groups (columns).
Groups: Vertical columns (e.g., Alkali Metals, Alkaline Earth Metals, Halogens, Noble Gases).
Periods: Horizontal rows.
Metals: High luster, good conductors, malleable.
Nonmetals: Poor conductors, dull appearance.
Metalloids: Properties intermediate between metals and nonmetals.

Atomic Structure
Atoms are composed of subatomic particles: protons, neutrons, and electrons.
Proton: Positive charge, mass = 1 amu, located in nucleus.
Neutron: No charge, mass = 1 amu, located in nucleus.
Electron: Negative charge, mass ≈ 0 amu, located outside nucleus.
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons and different mass numbers. Isotopes have identical chemical behavior but different stability.
Electronic Arrangement in Atoms
Electrons are arranged in energy levels (shells) around the nucleus. The maximum number of electrons in each shell is given by , where is the shell number.
Valence Shell: Outermost shell/highest energy level.
Valence Electrons: Electrons in the valence shell, important for chemical reactivity.
Nuclear Chemistry
Radioactivity and Nuclear Reactions
Nuclear chemistry studies the properties and reactions occurring in the nucleus of an atom. Unstable nuclei emit particles and energy (radiation).
Alpha Decay: Release of an alpha particle (helium nucleus).
Beta Decay: Neutron becomes a proton, emits an electron.
Positron Emission: Proton turns into a neutron, emits a positron.
Gamma Radiation: High-energy electromagnetic radiation, often emitted with other decay types.
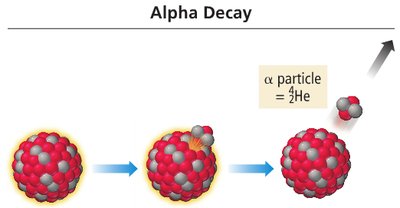
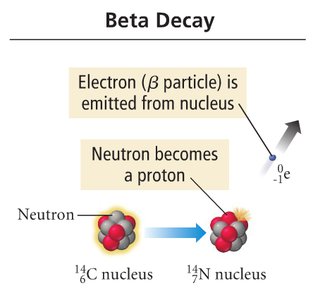
Balancing Nuclear Equations
Nuclear equations must conserve both mass number and atomic number.
Example:
Half-Life
The half-life () is the time required for half of a radioactive substance to decay. Each isotope has a characteristic half-life, which measures its stability.
Where is elapsed time and is the number of half-lives.
Ionic and Molecular Compounds
Chemical Nomenclature
Chemical formulas represent compounds. Ions are atoms or compounds with charges. The octet rule states that atoms with eight valence electrons are stable.
Cations: Positive ions formed by metals losing electrons.
Anions: Negative ions formed by nonmetals gaining electrons.
Polyatomic Ions: Groups of atoms with a charge.

Writing and Naming Ionic Compounds
Combine cation and anion to form a neutral compound.
Use parentheses for polyatomic ions when more than one is present.
Name cation first, anion second.
Use Roman numerals for variable charge metals.
Ionic Bond Formation
Ionic bonds form when ions of opposite charge attract each other, resulting in a compound that is neutral overall.
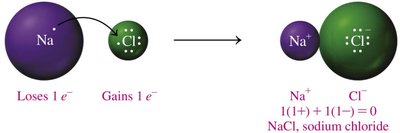
Naming Compounds Starting with Hydrogen
Compounds starting with hydrogen are named like ionic compounds (e.g., HCl).
Additional info:
Some explanations and context were expanded for completeness and clarity, including details on measurement precision, significant figures, and the scientific method. All images included are directly relevant to the adjacent content and reinforce key concepts.
