 Back
BackCHMY 123 Midterm Practice: Organic Chemistry Concepts and Nomenclature
Study Guide - Smart Notes

Q1. Which one of the following properties is a property of alkanes?
Background
Topic: Properties of Alkanes
This question tests your understanding of the physical and chemical properties of alkanes, a class of hydrocarbons with only single bonds.
Key Terms:
Alkanes: Saturated hydrocarbons with only C–C and C–H single bonds.
Melting point, density, solubility, reactivity, hydrogen bonding: Physical and chemical properties to compare.
Step-by-Step Guidance
Recall that alkanes are nonpolar molecules and generally insoluble in water.
Consider how the length of the carbon chain affects melting and boiling points.
Think about the density of alkanes compared to water.
Review whether alkanes form hydrogen bonds or are chemically reactive.
Try solving on your own before revealing the answer!
Q2. What is the parent name of the following structure using IUPAC rules?
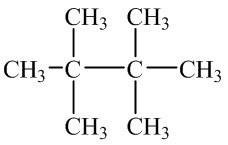
Background
Topic: IUPAC Nomenclature of Alkanes
This question tests your ability to identify the longest continuous carbon chain in a molecule and assign the correct parent name according to IUPAC rules.
Key Terms:
Parent chain: The longest continuous chain of carbon atoms.
IUPAC rules: Systematic method for naming organic compounds.
Step-by-Step Guidance
Identify all possible continuous carbon chains in the structure.
Count the number of carbons in each chain to find the longest one.
Assign the parent name based on the number of carbons (e.g., hexane for 6, octane for 8).
Try solving on your own before revealing the answer!
Q3. A carbon-carbon double bond of an alkene consists of:
Background
Topic: Bonding in Alkenes
This question tests your understanding of the types of bonds present in a carbon-carbon double bond, specifically sigma and pi bonds.
Key Terms and Formula:
Sigma (\sigma) bond: Formed by head-on overlap of orbitals.
Pi (\pi) bond: Formed by side-to-side overlap of p orbitals.
Step-by-Step Guidance
Recall that a single bond is a sigma bond.
Understand that a double bond consists of one sigma bond and one pi bond.
Visualize the orbital overlap to distinguish between sigma and pi bonds.
Try solving on your own before revealing the answer!
Q4. What is the IUPAC name of the compound shown?
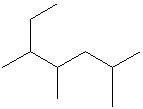
Background
Topic: IUPAC Nomenclature of Alkanes
This question tests your ability to name a branched alkane using IUPAC rules, including identifying substituents and their positions.
Key Terms:
Substituent: A group attached to the main carbon chain.
Numbering: Assigning numbers to the carbon chain to give substituents the lowest possible numbers.
Step-by-Step Guidance
Identify the longest carbon chain in the molecule.
Number the chain from the end closest to the first substituent.
List the substituents and their positions, then assemble the name in proper order.
Try solving on your own before revealing the answer!
Q5. How many secondary carbons are on the above molecule in question 4?
Background
Topic: Classification of Carbon Atoms
This question tests your ability to identify secondary carbons, which are carbons attached to two other carbons.
Key Terms:
Primary carbon: Attached to one other carbon.
Secondary carbon: Attached to two other carbons.
Tertiary carbon: Attached to three other carbons.
Quaternary carbon: Attached to four other carbons.
Step-by-Step Guidance
Examine each carbon in the molecule and count how many other carbons it is attached to.
Identify and count all secondary carbons.
Try solving on your own before revealing the answer!
Q6. Which of the following is an isomer of the molecule shown below?

Background
Topic: Structural Isomerism
This question tests your ability to recognize structural isomers—compounds with the same molecular formula but different connectivity.
Key Terms:
Isomer: Molecules with the same formula but different structures.
Structural isomer: Different connectivity of atoms.
Step-by-Step Guidance
Determine the molecular formula of the given molecule.
Compare the molecular formula of each option to see which matches.
Check the connectivity to ensure it is a structural isomer, not identical or unrelated.
Try solving on your own before revealing the answer!
