 Back
BackClassification and Balancing of Chemical Reactions (GOB Chemistry Ch.5 Study Guide)
Study Guide - Smart Notes

Classification and Balancing of Chemical Reactions
Concept: Chemical Reaction and Chemical Change
A chemical reaction is a process in which substances (reactants) undergo a chemical change to form new substances (products). This involves the breaking of chemical bonds in the reactants and the formation of new bonds in the products. Chemical reactions are a type of chemical change, distinct from physical changes, as they result in the formation of new substances with different properties.
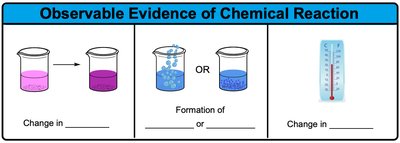
Evidence of a Chemical Reaction: Observable changes that indicate a chemical reaction has occurred include:
Change in color
Formation of a gas (bubbles)
Formation of a precipitate (solid)
Change in temperature (heat absorbed or released)
A chemical equation uses chemical formulas and symbols to represent a chemical reaction. For example, the decomposition of sodium bicarbonate can be written as:
Symbols used in chemical equations:
(s): solid
(l): liquid
(g): gas
(aq): aqueous (dissolved in water)
Δ: heat is applied
Law of Conservation of Mass
The Law of Conservation of Mass, established by Antoine Lavoisier, states that in a chemical reaction, matter is neither created nor destroyed. The total mass of reactants equals the total mass of products. In a chemical equation, substances before the arrow are reactants and those after are products.
All reactants are converted to products with nothing lost.
Mass is conserved:
Example: If 25.0 g of hydrogen reacts with 12.0 g of oxygen, the mass of water vapor formed is:
Mass of water vapor = 25.0 g + 12.0 g = 37.0 g
Balancing Chemical Equations
Balancing chemical equations ensures the same number and type of atoms are present on both sides of the equation, in accordance with the Law of Conservation of Mass. The numbers in front of formulas are called coefficients.
List elements in reactants and products.
Count atoms of each element on both sides.
Treat polyatomic ions as a unit if they appear unchanged on both sides.
Adjust coefficients to balance each element.
If necessary, multiply the entire equation to clear fractions.
Example: Balance the equation:
Solubility Rules
Solubility and Precipitation
Solubility is the ability of a solute to dissolve in a solvent. A soluble compound dissolves in water, while an insoluble compound does not. The solubility rules help predict whether an ionic compound will dissolve in water or form a precipitate (solid).
Soluble Compounds (GANA CASH)
GANA CASH is a mnemonic for common soluble ions:
G: Group 1 metals (Li+, Na+, K+)
A: Acetate (C2H3O2-)
N: Nitrate (NO3-)
A: Ammonium (NH4+)
C: Chlorate (ClO3-), Perchlorate (ClO4-)
A: None
S: Sulfate (SO42-)
H: Halides (F-, Cl-, Br-, I-)
Exceptions: Some ions form insoluble compounds with CBS (Ca2+, Ba2+, Sr2+) or HAP (Hg22+, Ag+, Pb2+).

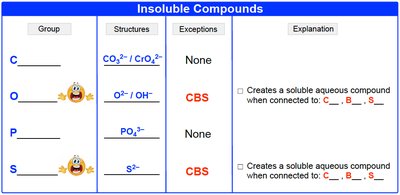
Insoluble Compounds (COPS)
COPS is a mnemonic for common insoluble ions:
C: Carbonate (CO32-), Chromate (CrO42-)
O: Oxide (O2-), Hydroxide (OH-)
P: Phosphate (PO43-)
S: Sulfide (S2-)
Exceptions: Some become soluble with CBS (Ca2+, Ba2+, Sr2+).
Molecular, Complete Ionic, and Net Ionic Equations
Molecular Equations
A molecular equation shows all reactants and products as intact compounds. Types include:
Neutralization: Acid + Base → Salt + Water
Gas Evolution: Reactants → Gas + Other Products
Precipitation: Reactants → At least one insoluble product (precipitate)
To write a molecular equation:
Break reactants into ions.
Swap ionic partners (combine cations with new anions).
Check if a precipitate, gas, or water forms (use solubility rules).
Balance the equation.
Complete and Net Ionic Equations
Complete ionic equation: Shows all soluble ionic compounds as dissociated ions.
Net ionic equation: Shows only the ions and molecules directly involved in the reaction (removes spectator ions).
Write the balanced molecular equation.
Break up only aqueous compounds into ions.
Cancel out spectator ions to get the net ionic equation.
Oxidation Numbers and Redox Reactions
Oxidation Numbers
The oxidation number (or state) is a value assigned to an atom to indicate its degree of oxidation or reduction. For an atom in its natural state, the oxidation number is zero. For monoatomic ions, it equals the ion's charge.
Rules exist for assigning oxidation numbers in compounds.
For non-listed elements, treat as unknown and solve using the sum of oxidation numbers equal to the compound's charge.
Redox Reactions
Redox reactions involve the transfer of electrons between reactants. Oxidation is the loss of electrons (increase in oxidation number), and reduction is the gain of electrons (decrease in oxidation number).
Oxidizing agent: The substance that is reduced (gains electrons).
Reducing agent: The substance that is oxidized (loses electrons).
Mnemonic: LEO the lion says GER (Loss of Electrons is Oxidation; Gain of Electrons is Reduction).

Balancing Redox Reactions
Acidic Solutions
Balancing redox reactions in acidic solutions involves:
Split the reaction into two half-reactions (oxidation and reduction).
Balance all elements except H and O.
Balance O by adding H2O.
Balance H by adding H+.
Balance charge by adding electrons to the more positive side.
Multiply half-reactions to equalize electrons, then add and cancel intermediates.
Basic Solutions
Balancing in basic solutions follows the same steps as acidic, with an additional step:
After balancing as if in acid, add OH- to both sides to neutralize H+ and form H2O.
Cancel any water molecules that appear on both sides.
Summary Table: Solubility Rules (GANA CASH & COPS)
Mnemonic | Ion/Compound | Solubility | Exceptions |
|---|---|---|---|
GANA CASH | Group 1, Acetate, Nitrate, Ammonium, Chlorate, Sulfate, Halides | Soluble | CBS, HAP |
COPS | Carbonate, Oxide, Phosphate, Sulfide | Insoluble | CBS |
Additional info: CBS = Ca2+, Ba2+, Sr2+; HAP = Hg22+, Ag+, Pb2+
