 Back
BackClassification and Balancing of Chemical Reactions: Study Notes for General, Organic, and Biological Chemistry
Study Guide - Smart Notes

Classification and Balancing of Chemical Reactions
Chemical Equations
Chemical equations are symbolic representations of chemical reactions, showing the reactants and products involved. The law of conservation of mass states that mass is neither created nor destroyed in a chemical reaction, so the same number and type of atoms must appear on both sides of the equation.
Reactants: Substances that undergo change, written on the left side of the arrow.
Products: Substances formed, written on the right side of the arrow.
Conditions: Necessary conditions (e.g., heat) are written above the arrow.
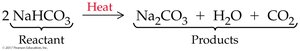
Balancing Chemical Equations
Balancing chemical equations ensures the conservation of mass by making sure the number and kinds of atoms are equal on both sides. Coefficients are placed in front of formulas to balance equations.
Coefficient: A number placed in front of a formula to balance the equation.
Subscripts in chemical formulas cannot be changed.
Balance elements that appear in only one compound first; leave elemental forms for last.
If a polyatomic ion appears on both sides, treat it as a single unit.
Check that coefficients are reduced to their lowest whole-number values.
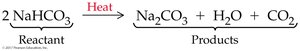
Law of Conservation of Mass
The law of conservation of mass states that the total mass of reactants equals the total mass of products. Atoms rearrange but are not created or destroyed.
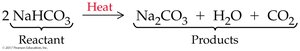
Phases or States in Chemical Equations
Physical states are indicated by symbols:
(s) = solid
(l) = liquid
(g) = gas
(aq) = dissolved in aqueous solution
Steps for Balancing Chemical Equations
Write an unbalanced equation using correct formulas for all reactants and products.
Add appropriate coefficients to balance the numbers of atoms of each element.
Check the equation to ensure the numbers and kinds of atoms are the same on both sides.
Reduce coefficients to their lowest whole-number values.
Types of Chemical Reactions
Precipitation Reactions
Precipitation reactions occur when two soluble ionic reactants produce an insoluble solid (precipitate) in aqueous solution.
Example: Ba(NO3)2 (aq) + K2CrO4 (aq) → BaCrO4 (s) + 2 KNO3 (aq)
Metal cations or ammonium ions switch partners.

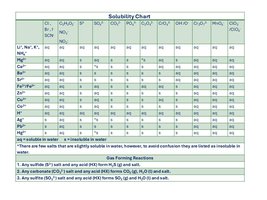
Solubility Guidelines
Solubility rules help predict whether a precipitation reaction will occur. Solubility is the amount of a compound that dissolves in a given solvent at a given temperature.
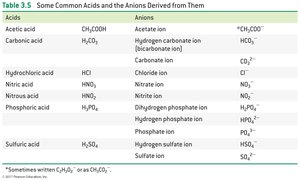
Acid-Base Neutralization Reactions
Acid-base neutralization reactions involve an acid reacting with a base to yield water and a salt. The H+ ion from the acid combines with the OH– ion from the base to form water, and the remaining ions form a salt.
Example: HCl (aq) + NaOH (aq) → H2O (l) + NaCl (aq)
Acids have H in front of an anion; bases are group 1 or 2 metals plus OH–.
Neutralization with Carbonates
Acids can react with carbonates or bicarbonates to yield water, a salt, and carbon dioxide.
General equation:
M = metal, HA = acid
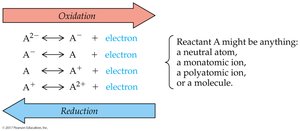
Oxidation-Reduction (Redox) Reactions
Redox reactions involve the transfer of electrons from one atom to another. Oxidation is the loss of electrons or increase in oxygen; reduction is the gain of electrons or decrease in oxygen.
Example: Zn (s) + 2 HCl (aq) → ZnCl2 (aq) + H2 (g)
Redox reactions are essential in batteries, combustion, respiration, bleaching, and metallurgy.
Redox Reaction Mechanism
Electron transfer changes the oxidation state of atoms. Oxidation increases charge or oxygen; reduction decreases charge or oxygen.
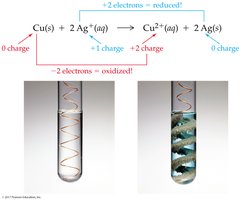
Redox Reaction Examples
Copper and Silver: Copper is oxidized, silver ions are reduced.
Iodide and Bromine: Iodide ion is oxidized, bromine is reduced.
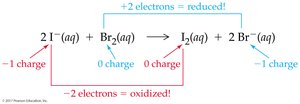
Reducing and Oxidizing Agents
Reducing agent: Loses electrons, causes reduction, undergoes oxidation.
Oxidizing agent: Gains electrons, causes oxidation, undergoes reduction.
Redox Reactions: Group I and II Metals
Alkali and alkaline earth metals are powerful reducing agents due to low ionization energy. As ionization energy decreases, reducing power increases.
Redox Reactions: Nonmetals
Reactive nonmetals are powerful oxidizing agents due to high electron affinity and electronegativity.
Recognizing Redox Reactions
Oxidation Numbers
Oxidation numbers indicate whether an atom is neutral, electron-rich, or electron-poor. They help track electron transfer in reactions.
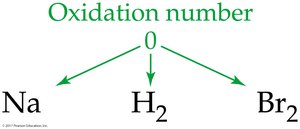
An atom in its elemental state has an oxidation number of 0.
A monatomic ion has an oxidation number equal to its charge.
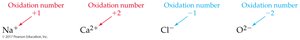
In molecular compounds, atoms usually have the same oxidation number as their monatomic ion.
Hydrogen is usually +1, oxygen is usually –2, nitrogen is usually –3, halogens are usually –1.
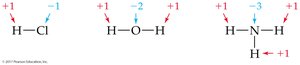
In compounds with more than one nonmetal, the more electronegative element has a negative oxidation number.
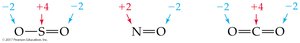
The sum of oxidation numbers in a neutral compound is 0.
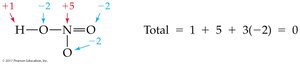
Net Ionic Equations
Definition and Writing Net Ionic Equations
Net ionic equations show only the ions and molecules directly involved in forming a liquid, gas, or solid. Spectator ions are omitted. Net ionic equations must be balanced for both atoms and charge.
All insoluble and molecular compounds are represented by their full formulas.
Liquids, gases, and solids are not separated into ions.
Examples of Net Ionic Equations
Precipitation: 2 AgNO3 (aq) + Na2CO3 (aq) → Ag2CO3 (s) + 2 NaNO3 (aq)
Neutralization: KOH (aq) + HNO3 (aq) → H2O (l) + KNO3 (aq)
Redox: Cu (s) + 2 AgNO3 (aq) → 2 Ag (s) + Cu(NO3)2 (aq)
Summary Table: Types of Chemical Reactions
Type | Description | Example |
|---|---|---|
Precipitation | Two soluble ionic reactants produce an insoluble solid | Ba(NO3)2 (aq) + K2CrO4 (aq) → BaCrO4 (s) + 2 KNO3 (aq) |
Neutralization | Acid reacts with base to produce water and salt | HCl (aq) + NaOH (aq) → H2O (l) + NaCl (aq) |
Redox | Electrons are transferred between atoms | Zn (s) + 2 HCl (aq) → ZnCl2 (aq) + H2 (g) |
Additional info: These notes expand on the original slides by providing definitions, stepwise procedures, and examples for each reaction type, as well as detailed explanations of oxidation numbers and net ionic equations. The included images directly reinforce key concepts and procedures.
