 Back
BackClassification of Matter and Chemical Reactions: Study Notes
Study Guide - Smart Notes

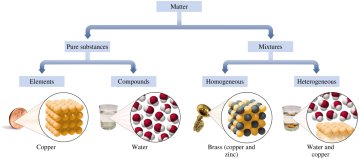
Classification of Matter
Definition and Overview
Matter is defined as anything that has mass and occupies space. All physical substances in the universe are forms of matter, which can be classified based on their composition and properties.
Pure substances have a fixed or definite composition.
Mixtures contain two or more different substances that are physically mixed but not chemically combined.
Pure Substances: Elements and Compounds
A pure substance is a type of matter with a fixed or definite composition. Pure substances are further classified as elements or compounds:
Elements are composed of only one type of atom (e.g., copper, Cu; lead, Pb; aluminum, Al).
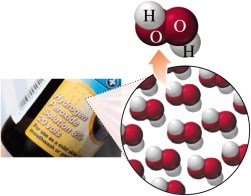
Compounds are composed of two or more elements combined in a fixed ratio (e.g., water, H2O; hydrogen peroxide, H2O2; table salt, NaCl).
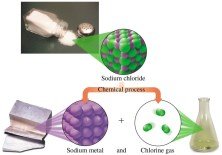
Elements in a Compound
Compounds can be broken down into their constituent elements by chemical processes. For example, sodium chloride (table salt) can be decomposed into sodium metal and chlorine gas.
Mixtures: Homogeneous and Heterogeneous
Mixtures consist of two or more substances physically combined. They can be separated by physical methods and may have variable composition.
Homogeneous mixtures (solutions) have a uniform composition throughout; the different parts are not visible (e.g., brass, a mixture of copper and zinc).
Heterogeneous mixtures have a non-uniform composition; the different parts are visible (e.g., a mixture of copper metal and water).

States and Properties of Matter
States of Matter
Matter exists in three primary states: solid, liquid, and gas. Each state is characterized by the arrangement and movement of its particles.
Solids: Definite shape and volume; particles are closely packed in a fixed arrangement and move very slowly.
Liquids: Indefinite shape but definite volume; particles are close together but can move past one another.
Gases: Indefinite shape and volume; particles are far apart and move rapidly.

Physical Properties
Physical properties are characteristics that can be observed or measured without changing the identity of a substance. Examples include shape, state, boiling and melting points, density, and color.
Example: Copper is reddish-orange, shiny, an excellent conductor of heat and electricity, solid at 25°C, with a melting point of 1083°C and boiling point of 2567°C.

Physical and Chemical Changes
A physical change alters the state or appearance of matter without changing its composition (e.g., melting, boiling, dissolving). A chemical change results in the formation of one or more new substances with different properties and composition (e.g., burning, rusting, caramelizing sugar).
Physical change: Water boiling, sugar dissolving, paper cutting.
Chemical change: Wood burning, iron rusting, sugar caramelizing.

Chemical Reactions and Equations
Evidence of Chemical Change
Chemical changes can be identified by several types of evidence:
Change in color
Formation of a gas (bubbles)
Formation of a solid (precipitate)
Heat or light produced or absorbed





Writing and Balancing Chemical Equations
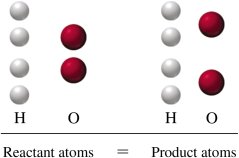
Chemical equations represent the substances involved in a chemical reaction. The reactants are written on the left, the products on the right, separated by an arrow. Equations must be balanced so that the number of atoms of each element is the same on both sides.
Symbols: (s) = solid, (l) = liquid, (g) = gas, (aq) = aqueous, Δ = heat
Example equation:

Types of Chemical Reactions
Chemical reactions are classified into several types based on the rearrangement of atoms:
Combination (Synthesis) Reaction: Two or more substances combine to form one product.
Decomposition Reaction: One substance splits into two or more simpler substances.
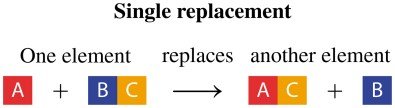
Single Replacement Reaction: One element replaces another in a compound.
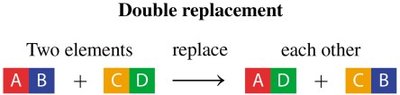
Double Replacement Reaction: The positive ions in two compounds exchange places.
Combustion Reaction: A carbon-containing compound burns in oxygen to produce CO2, H2O, and energy.


Summary Table: Types of Chemical Reactions
Reaction Type | General Equation | Example |
|---|---|---|
Combination | ||
Decomposition | ||
Single Replacement | ||
Double Replacement | ||
Combustion |
Additional info: These classifications and examples provide a foundation for understanding chemical changes and the conservation of mass in chemical reactions, which are essential concepts in introductory chemistry courses.
