 Back
BackChapter 3: Compounds
Study Guide - Smart Notes

Compounds and Electron Arrangements
Electron Cloud and Energy Levels
Electrons in an atom move around the nucleus in a region called the electron cloud. These electrons occupy distinct energy levels (quantified by the principal quantum number, n), which are determined by their energy. Electrons fill the lowest available energy levels first, starting with n=1 closest to the nucleus. The maximum number of electrons in a given energy level is calculated by the formula .
n = 1 holds 2 electrons
n = 2 holds 8 electrons
n = 3 holds 18 electrons
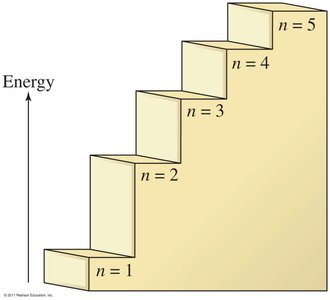
Energy-Level Arrangements of Electrons
The arrangement of electrons in energy levels determines the chemical properties of elements. The number of valence electrons (those in the outermost energy level) is especially important for reactivity.
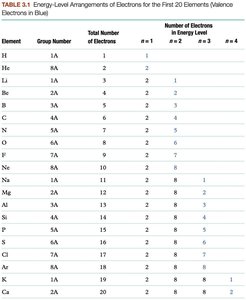
Periodic Table and Electron Arrangements
The group number in the periodic table indicates the number of valence electrons for main-group elements, while the period number indicates the outermost energy level containing valence electrons.
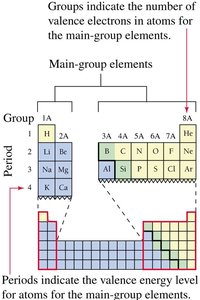
Valence Electrons and Chemical Bonds
Valence and Core Electrons
Valence electrons are found in the highest energy level and are responsible for chemical reactivity. Core electrons are all other electrons not involved in chemical reactions. (also the group number on the periodic table)
Electron-Dot (Lewis) Symbols
Lewis symbols represent the nucleus and core electrons with the element symbol, and valence electrons as dots around it. The first four dots are placed singly, then paired as needed.
blue = metals green = metalloid yellow = nonmetal
Octet Rule
Atoms are most stable when their valence shell is full, typically with eight electrons (an octet). Atoms will gain, lose, or share electrons to achieve an octet, except for helium, which is stable with two electrons (a duet).
Types of Chemical Bonds
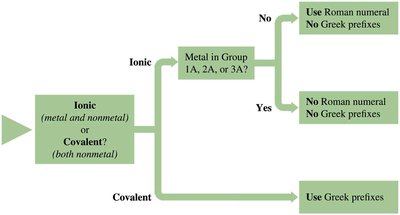
Ionic and Covalent Bonds
Atoms form bonds to achieve stability:
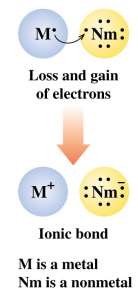
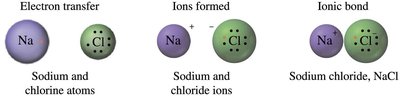
Ionic bonds: Formed between metals and nonmetals by transfer of electrons.
Covalent bonds: Formed between nonmetals by sharing electrons.
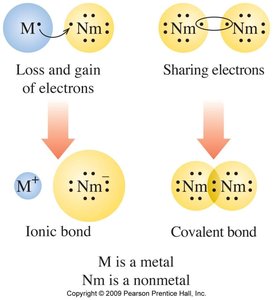
Ions: Cations and Anions
Ions are charged particles formed when atoms gain or lose electrons:
Cations: Positively charged, formed by loss of electrons (usually metals).
Anions: Negatively charged, formed by the gain of electrons (usually nonmetals).
Protons are never gained or lost.
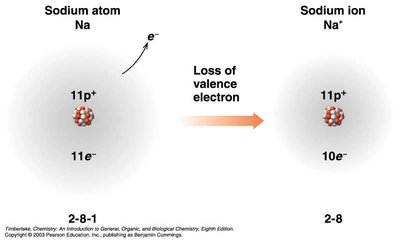
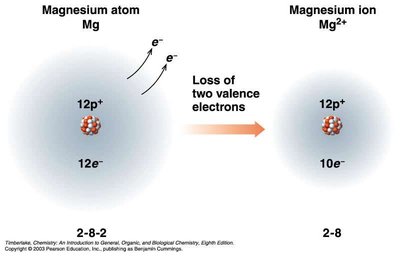
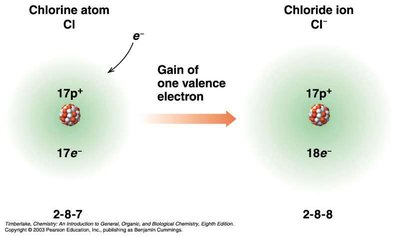
Common Charges on Monatomic Ions
Group numbers predict ion charges:
Group 1A: +1
Group 2A: +2
Group 3A: +3
Group 5A: -3
Group 6A: -2
Group 7A: -1
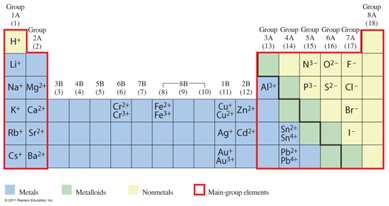
Transition metals (found in the center of the periodic table) can form more than one cation. Exceptions are Zn 2+, Cd2+ and Ag+ (always have these fixed charges)
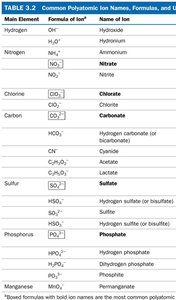
Polyatomic Ions
Polyatomic ions are molecules that have gained or lost electrons, resulting in a net charge. Examples include ammonium (NH4+), nitrate (NO3-), sulfate (SO42-), hydroxide (OH-), and carbonate (CO32-).
Naming Ionic and Covalent Compounds
Naming Monatomic Ions
Cations: Add "ion" to the metal name (e.g., Na+ is sodium ion).
Transition metals: Use Roman numerals to indicate charge (e.g., Fe2+ is iron(II) ion).
Anions: Replace the ending with "-ide" and add "ion" (e.g., Cl- is chloride ion).
Naming Polyatomic Ions
Polyatomic ions have unique names and formulas that must be memorized.

Ionic Compounds
Properties and Formation
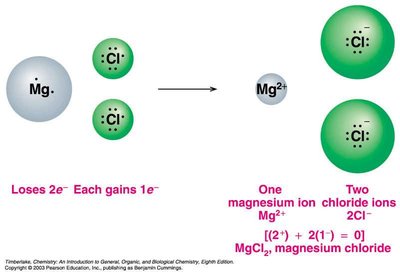
Ionic compounds are composed of positive and negative ions, held together by strong electrostatic forces (ionic bonds). They are electrically neutral, have high melting and boiling points, and are solid at room temperature.

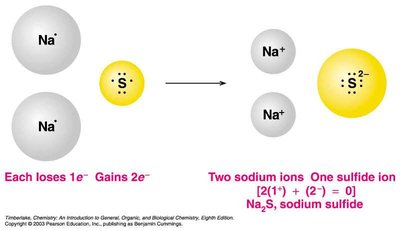
Writing Formulas and Names
Split the compound into cation and anion.
Write the name of the cation, then the anion.
Make the positive and negative charges equal when writing formulas.
Covalent Compounds
Covalent Bond Formation
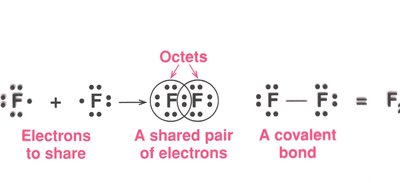
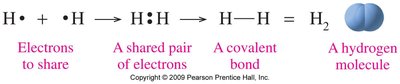
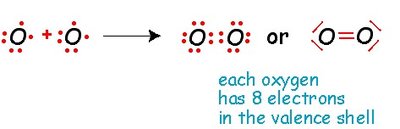
Covalent bonds are formed by the sharing of pairs of electrons between two nonmetals. The smallest unit of a covalent compound is a molecule.
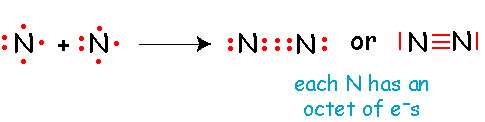
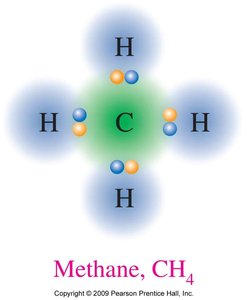
 Carbon is the most important
Carbon is the most important 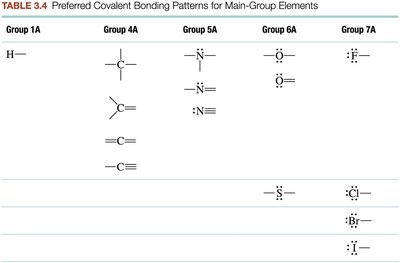
Bond Types and Lewis Structures
Single bond: One pair of electrons shared (All halogens)
Double bond: Two pairs of electrons shared
Triple bond: Three pairs of electrons shared
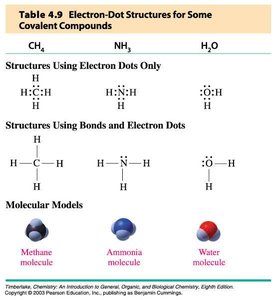
Writing Lewis Structures for Covalent Molecules
Count the total number of valence electrons.
Write a plausible skeletal structure. The element that is written first in the formula is the central atom (unless it’s hydrogen).
Connect the terminal atoms to the central atoms by single dashes (covalent bond = 1 pair of electrons).
Place pairs of electrons as lone pairs around the terminal atoms to give each terminal atom (except H) an octet.
Assign any remaining electrons as lone pairs around the central atom.
If necessary, move one or more lone pairs of electrons
Naming Covalent Compounds
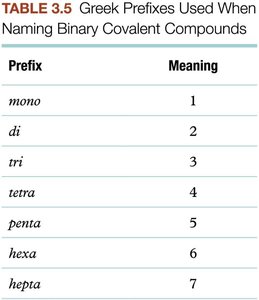
Binary molecular compounds (two different nonmetals) use Greek prefixes to indicate the number of atoms. The more metallic element is named first, and the less metallic element second, with its ending changed to "-ide".
Identifying Ionic vs. Covalent Compounds
A compound is ionic if the first element is a metal or the polyatomic ion NH4+. It is covalent if the first element is a nonmetal.
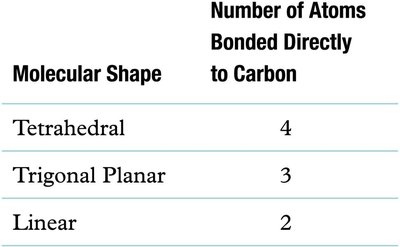
Shapes of Covalent Molecules
Molecular Geometry and VSEPR Theory
The function of molecules is determined by their three-dimensional shape, which is predicted by the Valence Shell Electron Pair Repulsion (VSEPR) model. Electron pairs arrange themselves as far apart as possible to minimize repulsion.
Linear: 180° bond angle
Planar triangle: 120° bond angle
Tetrahedral: 109.5° bond angle


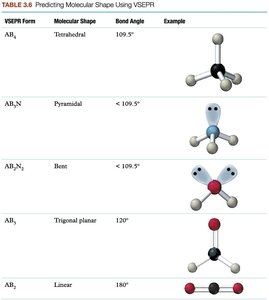
VSEPR Theory Table
Pairs of e- | Lone Pairs | Shape |
|---|---|---|
2 | 0 | Linear |
3 | 0 | Triangle |
3 | 1 | Bent |
4 | 0 | Tetrahedral |
4 | 1 | Pyramid |
4 | 2 | Bent |
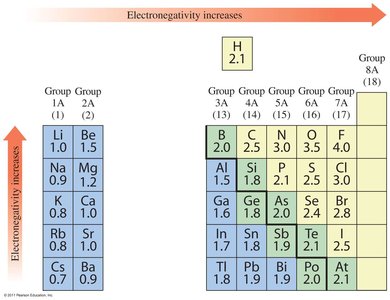
Electronegativity and Polarity
Electronegativity Trends
Electronegativity is the attraction an atom has for electrons in a bond. It increases across a period and decreases down a group. Fluorine is the most electronegative element.
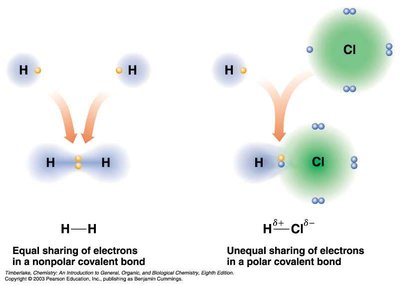
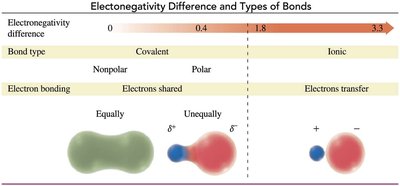
Bond Polarity
The greater the difference in electronegativity between two atoms, the more polar the bond. If the difference is large enough, the bond is ionic.
Polar and Nonpolar Molecules
A molecule is polar if it has polar bonds and its shape does not allow the polar bonds to cancel out. The rules are:
If the central atom has one or more pairs of nonbonding electrons, the molecule is polar.
If the central atom has no nonbonding pairs and all terminal atoms are identical, the molecule is nonpolar.
If the central atom has no nonbonding pairs and at least one terminal atom is different, the molecule is polar.

Counting Atoms and Moles
Counting Items: The Mole Concept
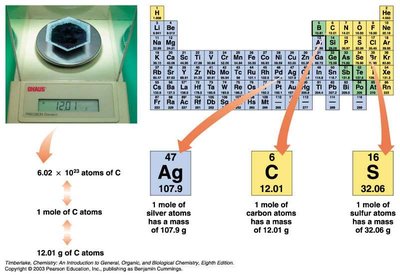
Chemists use the mole to relate the mass of an element in grams to the number of atoms it contains. One mole contains particles (Avogadro's number).

Avogadro's Number and Molar Mass
Avogadro's number () defines the number of atoms in one mole. The molar mass is the mass in grams of one mole of an element, numerically equal to its atomic mass.
Formula Mass and Molar Mass of Compounds
The formula mass is the sum of the atomic masses of the atoms in a compound. The molar mass is the mass of one mole of the compound, containing Avogadro's number of molecules.
For CH4: formula mass = 12.01 amu + 4(1.01 amu) = 16.05 amu/molecule
Molar mass of CH4 = 16.05 g/mole
Summary Table: Greek Prefixes for Covalent Compounds
Prefix | Meaning |
|---|---|
mono | 1 |
di | 2 |
tri | 3 |
tetra | 4 |
penta | 5 |
hexa | 6 |
hepta | 7 |
