 Back
BackCompounds—How Elements Combine: Covalent Bond Formation and The Mole
Study Guide - Smart Notes

Compounds—How Elements Combine
Covalent Bond Formation
Covalent bonds are formed when atoms share electrons to achieve a stable electron configuration, typically an octet. This section explores how nonmetals combine to form covalent compounds, the relationship between valence electrons and bonding, and the drawing of Lewis structures.
Covalent Bond: A chemical bond formed by the sharing of electron pairs between atoms.
Covalent Compound: A compound composed of molecules formed by covalent bonds.
Molecule: The smallest unit of a covalent compound that retains its chemical properties.
Octet Rule: Atoms tend to form bonds until they are surrounded by eight valence electrons.
Bonding Capacity: The number of covalent bonds an atom forms equals the number of electrons needed to complete its octet.
Examples: Carbon forms four bonds, nitrogen three, oxygen two, and halogens (like fluorine and chlorine) one.

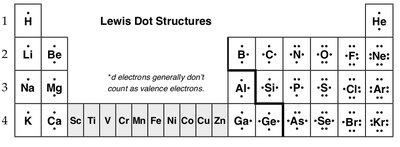
Lewis Dot Structures
Lewis dot structures visually represent the valence electrons of atoms and help predict bonding patterns in molecules. Each dot represents a valence electron, and the arrangement of dots shows possible bonding sites.
Electron-dot Symbol: The elemental symbol surrounded by dots representing valence electrons.
Drawing Lewis Structures: Steps include drawing electron-dot symbols, arranging atoms, connecting with bonds, and confirming complete valence shells.
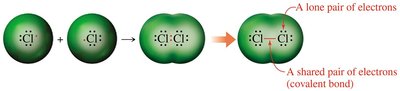
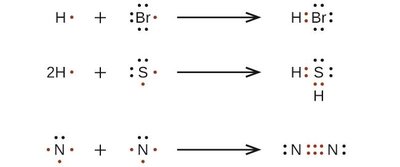
Bonding Patterns in Covalent Compounds
The number and type of bonds formed by an atom depend on its valence electrons. Carbon, for example, can form single, double, or triple bonds to complete its octet.
Single Bond: Sharing one pair of electrons.
Double Bond: Sharing two pairs of electrons.
Triple Bond: Sharing three pairs of electrons.
Example: Carbon in methane (CH4) forms four single bonds.
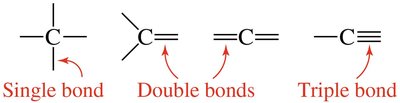
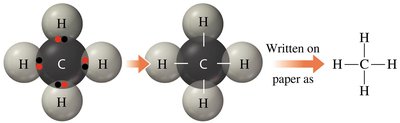
Formulas and Structures of Covalent Compounds
Covalent compounds are represented by molecular formulas indicating the number and type of atoms present. Lewis structures provide a visual representation of bonding and electron arrangement.
Example: Glucose (C6H12O6) contains 6 carbon, 12 hydrogen, and 6 oxygen atoms.
Steps for Drawing Lewis Structures:
Draw electron-dot symbols for each atom.
Arrange atoms to maximize bonding.
Connect atoms with bonds (lines).
Ensure all atoms have complete valence shells.
Check the structure for accuracy.
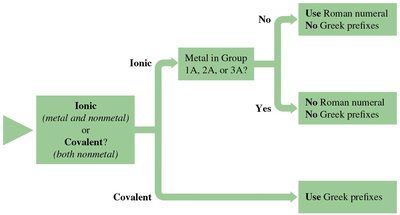
Naming Binary Covalent Compounds
Binary covalent compounds consist of two nonmetal elements. Their names follow a systematic procedure using Greek prefixes to indicate the number of each atom.
Step 1: Name the first element in the formula.
Step 2: Name the second element, changing the ending to -ide.
Step 3: Use Greek prefixes to indicate the number of each element (mono-, di-, tri-, tetra-, etc.).
Note: The prefix 'mono-' is only used for the second element.
Examples: CO (carbon monoxide), NO2 (nitrogen dioxide), N2O4 (dinitrogen tetroxide).
Prefix | Meaning |
|---|---|
mono- | 1 |
di- | 2 |
tri- | 3 |
tetra- | 4 |
penta- | 5 |
hexa- | 6 |
hepta- | 7 |
The Mole: Counting Atoms and Compounds
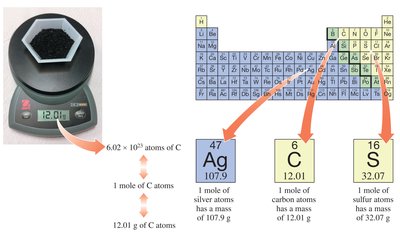
The Mole and Avogadro’s Number
The mole is a fundamental unit in chemistry that relates the mass of a substance to the number of particles it contains. Avogadro’s number defines the number of atoms or molecules in one mole.
Mole (mol): The amount of substance containing particles (atoms, molecules, or ions).
Avogadro’s Number (N): particles per mole.
Conversion: Use Avogadro’s number to convert between moles and number of particles.
Example: 2.5 moles of sodium contains atoms.
Molar Mass and Formula Weight
Molar mass is the mass of one mole of a substance, numerically equal to the sum of the atomic masses of its constituent atoms (formula weight). The dalton (Da) is used for large molecules, especially in biochemistry.
Formula Weight: The sum of atomic masses in a compound (in amu).
Molar Mass: The mass of one mole of a compound (in grams/mole).
Dalton (Da): Equivalent to the atomic mass unit (amu), used for large biomolecules.
Example Calculation:
Propane (C3H8): Calculate molar mass by adding atomic masses of all atoms.
Mass of 0.25 mol of propane: Multiply molar mass by 0.25.
Key Equations:
Mole to atoms:
Mole to mass:
Mass to moles:
