 Back
BackCompounds—How Elements Combine: Electron Arrangements, Ion Formation, and Ionic Compounds
Study Guide - Smart Notes

Chapter 3: Compounds—How Elements Combine
3.1 Electron Arrangements and the Octet Rule
Understanding how electrons are arranged in atoms is fundamental to predicting chemical behavior. The arrangement of electrons in shells (energy levels) determines the chemical properties of elements, especially for the main-group elements in the first four periods of the periodic table.
Electron Shells: Electrons occupy discrete energy levels (shells) around the nucleus, labeled by the principal quantum number n (e.g., n = 1, 2, 3, 4).
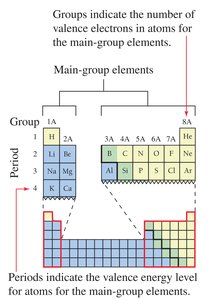
Valence Electrons: The electrons in the outermost shell are called valence electrons. These are responsible for chemical bonding.
Periodic Table Groups: Elements in the same group have the same number of valence electrons, which explains their similar chemical properties.
Octet Rule: Most atoms tend to react to achieve eight electrons in their valence shell, known as the octet rule. Noble gases (Group 8A) are stable because they naturally possess a full valence shell.
Exceptions: Helium is stable with only two valence electrons.
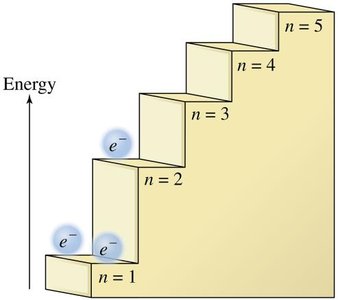
Example: Sodium (Na) has one valence electron in the third energy level (n = 3), while neon (Ne) has eight valence electrons in the second energy level (n = 2).
3.2 In Search of an Octet, Part 1: Ion Formation
Atoms form ions to achieve a stable electron configuration, often following the octet rule. Ion formation involves the loss or gain of electrons, resulting in charged species.
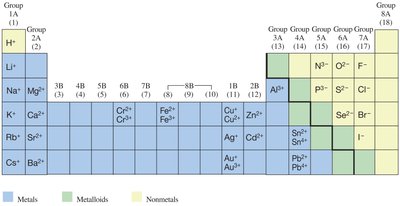
Cations: Positively charged ions formed when an atom loses one or more electrons. Typically formed by metals (e.g., Na+, Ca2+).
Anions: Negatively charged ions formed when an atom gains one or more electrons. Typically formed by nonmetals (e.g., Cl-, O2-).
Isoelectronic: Ions that have the same electron configuration as a noble gas.
Polyatomic Ions: Groups of covalently bonded atoms that carry a net charge (e.g., SO42-, NH4+).
Ion Charges: The charge of an ion can be predicted from its group number in the periodic table for main-group elements.
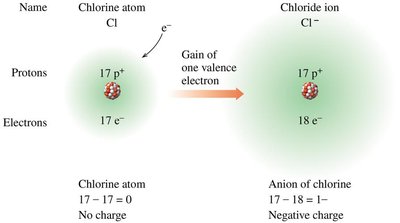
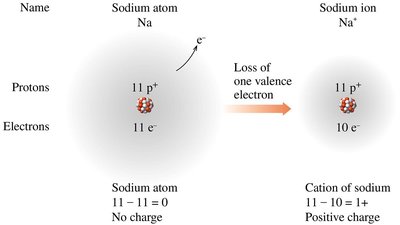
Example: Sodium (Na) loses one electron to form Na+; chlorine (Cl) gains one electron to form Cl-.
Naming Ions
Metal Ions: Add the word "ion" to the metal name (e.g., sodium ion).
Transition Metals: Use Roman numerals to indicate charge (e.g., iron(II) ion for Fe2+).
Nonmetal Ions: Replace the ending with "-ide" (e.g., chloride).
Polyatomic Ions: Most end in "-ate" or "-ite" (e.g., nitrate, nitrite). Exceptions include hydroxide (OH-), hydronium (H3O+), and cyanide (CN-).
Common Biological Ions and Their Functions
Cation | Function | Sources |
|---|---|---|
Na+ | Regulates fluids outside cells | Table salt, seafood |
K+ | Maintains ion concentration in cells; induces heartbeat | Dairy, bananas, meat |
Ca2+ | Muscle contraction, bone/teeth formation, heartbeat regulation | Dairy, whole grains, leafy vegetables |
Mg2+ | Nerve impulse transmission | Nuts, seafood, leafy vegetables |
Fe2+ | Oxygen transport in hemoglobin | Liver, red meat, leafy vegetables |
Anion | Function | Sources |
|---|---|---|
Cl- | Fluid balance, gastric juice | Table salt, seafood |
HCO3- | Acid–base balance in blood | Produced by body |
HPO42- | Acid–base balance in cells | Fish, poultry, dairy |
Common Polyatomic Ions
Main Element | Formula | Name | Common Uses |
|---|---|---|---|
Hydrogen | OH- | Hydroxide | Antacids |
Hydrogen | H3O+ | Hydronium | Measure of acidity (pH) |
Nitrogen | NH4+ | Ammonium | Indicator of liver/kidney malfunction |
Nitrogen | NO3- | Nitrate | Preservative |
Nitrogen | NO2- | Nitrite | Prevents botulism in meats |
Chlorine | ClO3- | Chlorate | Disinfectant |
Carbon | CO32- | Carbonate | Antacids, depression treatment |
Carbon | HCO3- | Bicarbonate | Acid–base balance in blood |
Carbon | CN- | Cyanide | Cellular respiration inhibitor |
Sulfur | SO42- | Sulfate | Reduces inflammation (Epsom salts) |
Phosphorus | PO43- | Phosphate | Energy transfer in body |
3.3 Ionic Compounds—Electron Give and Take
Ionic compounds are formed when metals and nonmetals combine through the transfer of electrons, resulting in the formation of cations and anions. The electrostatic attraction between these oppositely charged ions is called an ionic bond.
Formation: Metals lose electrons to form cations; nonmetals gain electrons to form anions.
Charge Balance: Ionic compounds are electrically neutral; the total positive and negative charges must balance.
Formulas: The chemical formula shows the ratio of ions needed to achieve neutrality. Subscripts indicate the number of each ion present.
Naming: The cation name is followed by the anion name (e.g., sodium chloride for NaCl). For transition metals, a Roman numeral indicates the charge (e.g., copper(II) oxide for CuO).
Polyatomic Ions: If present, the polyatomic ion name remains unchanged in the compound name (e.g., calcium phosphate for Ca3(PO4)2).
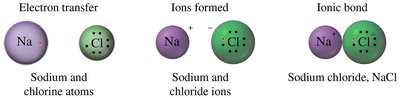
Examples:
Mg2+ and Cl- combine to form MgCl2.
Na+ and S2- combine to form Na2S.
Al3+ and O2- combine to form Al2O3.
Cu+ and CO32- combine to form Cu2CO3.
Fe2+ and NO3- combine to form Fe(NO3)2.
Key Equation:
Additional info: The process of forming ionic compounds is essential in biological systems, such as the formation of salts and electrolytes necessary for nerve transmission and fluid balance.
