 Back
BackCovalent Compounds: Structure, Bonding, and Properties
Study Guide - Smart Notes

Covalent Compounds: Structure, Bonding, and Properties
Introduction to Covalent Bonding
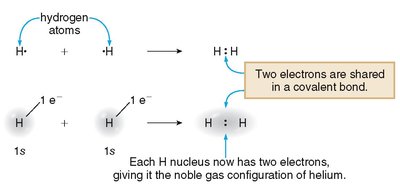
Covalent bonds are a fundamental type of chemical bond where two atoms share electrons to achieve a stable electron configuration. This sharing allows each atom to attain the electron arrangement of the nearest noble gas, resulting in the formation of discrete molecules.
Covalent bond: A chemical bond formed by the sharing of one or more pairs of electrons between two atoms.
Molecule: A group of atoms held together by covalent bonds, representing the smallest unit of a covalent compound.
Diatomic Elements
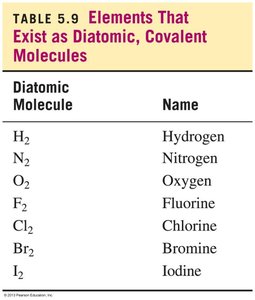
Certain elements naturally exist as diatomic molecules, meaning they are found in nature as pairs of atoms bonded together by covalent bonds. These elements are essential in both chemical reactions and biological processes.
The seven diatomic elements are: H2, N2, O2, F2, Cl2, Br2, I2.
Nomenclature of Covalent Compounds
Naming Covalent Compounds
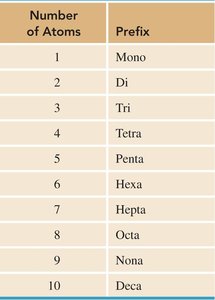
Covalent compounds are named using a systematic approach that indicates the number and type of atoms present. Prefixes are used to denote the number of each atom, and the second element's name ends with the suffix "-ide." The prefix "mono-" is usually omitted for the first element.
First nonmetal: Use the element name.
Second nonmetal: Use the root of the element name + "-ide".
Prefixes indicate the number of atoms (see table below).
Examples:
CO: carbon monoxide
NO2: nitrogen dioxide
N2O4: dinitrogen tetroxide
Writing Formulas for Covalent Compounds
To write the formula from a compound's name, use the element symbols in the order given and apply the appropriate prefixes as subscripts.
Example: Sulfur hexafluoride → SF6
Example: Dinitrogen trioxide → N2O3
Lewis Structures and Resonance
Lewis Structures
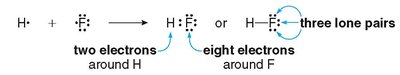
Lewis structures are diagrams that show the arrangement of valence electrons among atoms in a molecule. They help predict molecular shape, bond order, and the presence of lone pairs.
Each bond represents two shared electrons.
Lone pairs are non-bonding pairs of electrons localized on a single atom.
Atoms (except hydrogen) strive for an octet (eight electrons) in their valence shell.
Resonance Structures
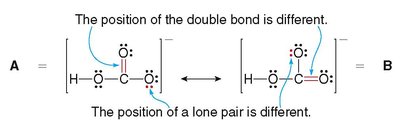
Some molecules cannot be adequately represented by a single Lewis structure. Resonance structures are two or more valid Lewis structures that differ only in the position of electrons, not atoms. The true structure is a resonance hybrid, which stabilizes the molecule by delocalizing electrons.
Resonance structures have the same arrangement of atoms but different arrangements of electrons.
The actual molecule is a hybrid of all resonance forms.
Molecular Shape and VSEPR Theory
VSEPR Theory
The Valence Shell Electron Pair Repulsion (VSEPR) theory predicts the three-dimensional shape of molecules based on the repulsion between electron groups (bonding and lone pairs) around a central atom. The arrangement minimizes repulsion, resulting in specific molecular geometries.
Electron groups arrange themselves as far apart as possible.
The number of electron groups determines the molecular geometry.
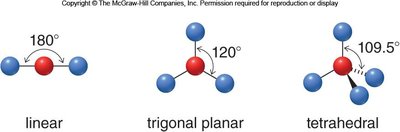
Common Molecular Geometries
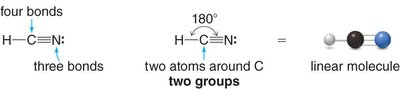
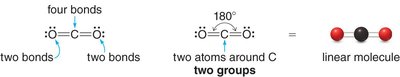
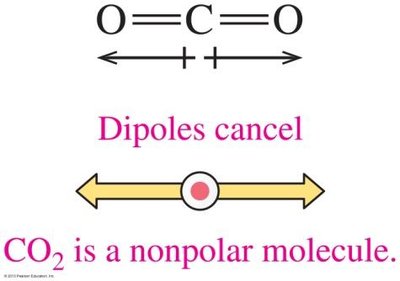
Linear: 2 groups, 180° bond angle (e.g., CO2)
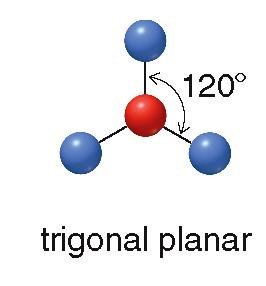
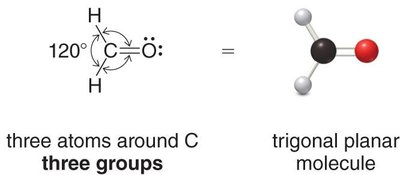
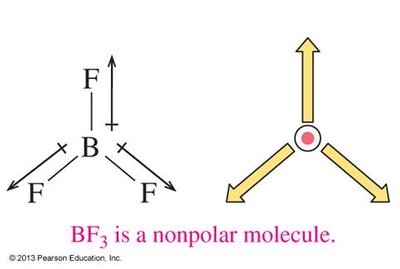
Trigonal planar: 3 groups, 120° bond angle (e.g., BF3)
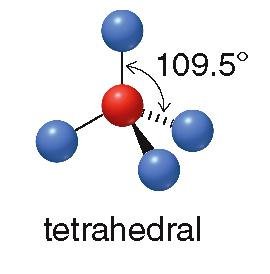
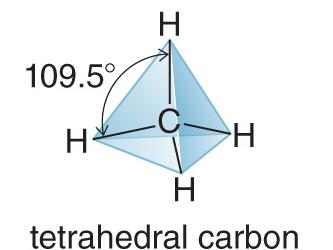
Tetrahedral: 4 groups, 109.5° bond angle (e.g., CH4)
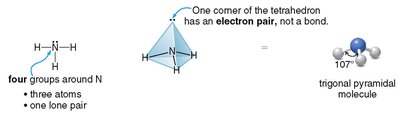
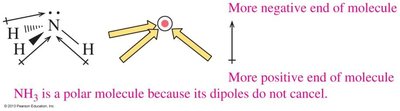
Trigonal pyramidal: 3 atoms + 1 lone pair (e.g., NH3), bond angle ~107°
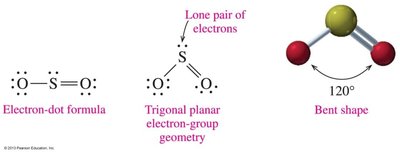
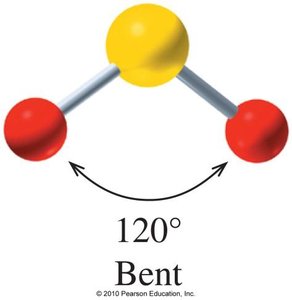
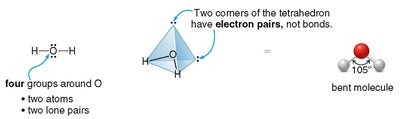
Bent: 2 atoms + 1 or 2 lone pairs (e.g., H2O, SO2), bond angle ~105° or 120°
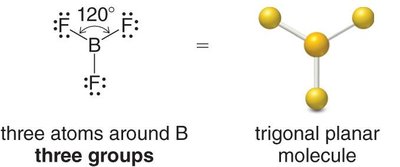
Electronegativity and Bond Polarity
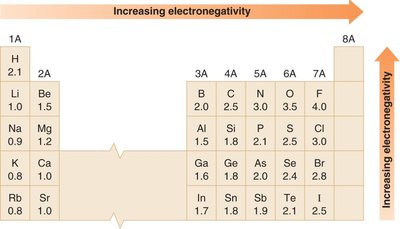
Electronegativity
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. It increases across a period and decreases down a group in the periodic table. Fluorine is the most electronegative element.
Bond Polarity
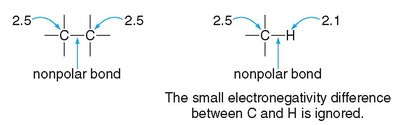
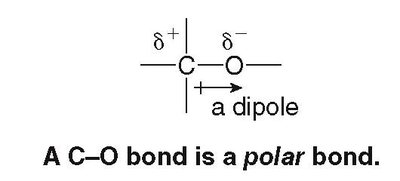
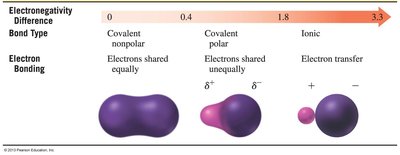
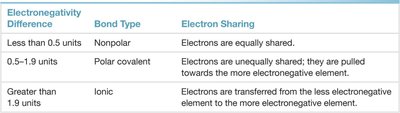
The difference in electronegativity between two bonded atoms determines the bond type:
Nonpolar covalent: Electrons are shared equally (ΔEN < 0.5).
Polar covalent: Electrons are shared unequally (0.5 ≤ ΔEN ≤ 1.9).
Ionic: Electrons are transferred (ΔEN > 1.9).
Polarity of Molecules
Determining Molecular Polarity
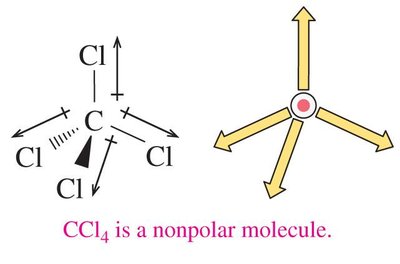
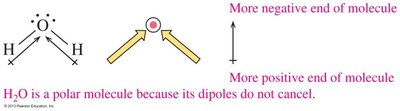
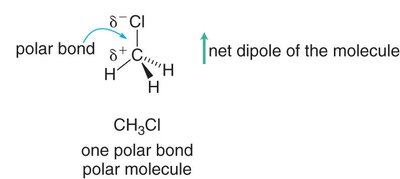
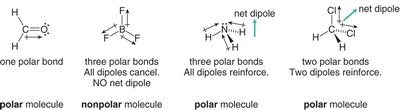
The overall polarity of a molecule depends on both the polarity of its bonds and its molecular geometry. If the dipoles cancel due to symmetry, the molecule is nonpolar; if they reinforce, the molecule is polar.
Linear, trigonal planar, and tetrahedral molecules with identical surrounding atoms are usually nonpolar.
Bent and trigonal pyramidal molecules are usually polar.
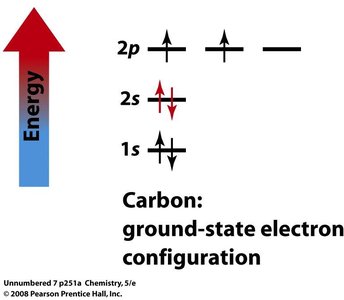
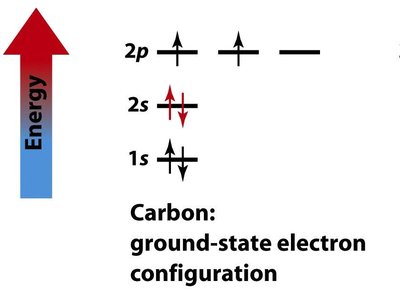
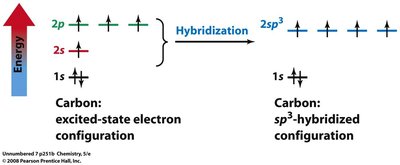
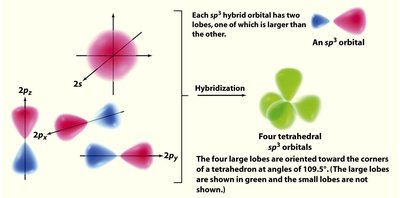
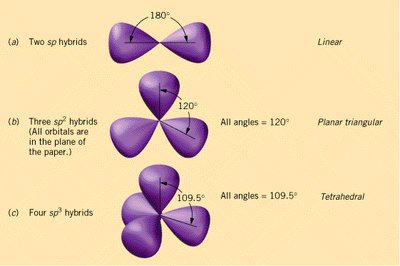
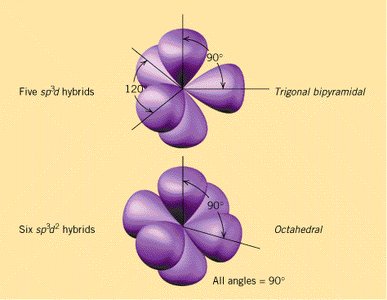
Hybridization
Hybridization and Molecular Geometry
Hybridization explains the observed shapes of molecules by describing the mixing of atomic orbitals to form new, equivalent hybrid orbitals. The type of hybridization corresponds to the number of electron groups around the central atom.
sp: 2 groups, linear geometry
sp2: 3 groups, trigonal planar geometry
sp3: 4 groups, tetrahedral geometry
Intermolecular Forces
Types of Intermolecular Forces (IMFs)
Intermolecular forces are attractions between molecules that influence physical properties such as boiling and melting points. They are generally weaker than intramolecular (covalent or ionic) bonds.
London Dispersion Forces (LDF): Present in all molecules; weakest IMF; arise from temporary dipoles.
Dipole-Dipole Forces: Present in polar molecules; due to permanent dipoles.
Hydrogen Bonding: Special dipole-dipole interaction involving H bonded to O, N, or F; stronger than regular dipole-dipole forces.
Ion-Dipole Forces: Attraction between an ion and a polar molecule; strongest IMF.
Periodic Trends
Effective Nuclear Charge (Zeff)
The effective nuclear charge is the net positive charge experienced by valence electrons, accounting for shielding by inner electrons. It increases across a period and decreases down a group.
Formula:
Atomic Size
Atomic size (radius) increases down a group and decreases across a period due to increasing nuclear charge pulling electrons closer.
Ionization Energy (IE)
Ionization energy is the energy required to remove an electron from a neutral atom. It decreases down a group and increases across a period.
Electron Affinity (EA)
Electron affinity is the energy released when an atom gains an electron. It generally becomes more negative (greater) across a period.
Metallic Character
Metallic character increases down a group and decreases across a period. Elements with high metallic character lose electrons easily.
*Additional info: This guide covers the essential concepts of covalent bonding, molecular structure, polarity, hybridization, intermolecular forces, and periodic trends, providing a comprehensive foundation for further study in general chemistry.*
