 Back
BackEnergy Diagrams and Reaction Types: Analyzing Exergonic and Endergonic Reactions
Study Guide - Smart Notes

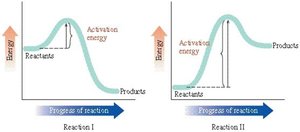
Q4. Determine which of the statements is incorrect regarding this figure:
Background
Topic: Reaction Energy Profiles (Exergonic vs. Endergonic Reactions)
This question tests your understanding of energy diagrams for chemical reactions, specifically the concepts of activation energy, exergonic and endergonic reactions, and how to interpret reaction progress graphs.
Key Terms and Concepts:
Activation Energy (): The minimum energy required for a reaction to occur, shown as the peak between reactants and products.
Exergonic Reaction: A reaction where products have lower free energy than reactants; energy is released.
Endergonic Reaction: A reaction where products have higher free energy than reactants; energy is absorbed.
Energy Profile Diagram: A graph plotting energy versus progress of reaction, showing reactants, products, and activation energy.
Step-by-Step Guidance
Examine the energy diagrams for both reactions. Notice the starting and ending points for reactants and products in each diagram.
Identify which reaction is exergonic and which is endergonic by comparing the energy levels of reactants and products:
For Reaction I, products are lower in energy than reactants (exergonic).
For Reaction II, products are higher in energy than reactants (endergonic).
Compare the activation energies for both reactions by looking at the height of the energy barrier (the peak) for each diagram.
Consider the statements given in the question and use the diagrams to evaluate their accuracy. Pay attention to which reaction is faster, which is exergonic/endergonic, and the relative activation energies.
Try solving on your own before revealing the answer!
Final Answer: A) Reaction II occurs faster than reaction I.
Reaction II has a higher activation energy than Reaction I, so it would actually occur slower, not faster. The other statements are correct based on the diagrams.
