 Back
BackEnergy Diagrams and Reaction Types, Carbohydrate Structures, and Glycosidic Bonds
Study Guide - Smart Notes

Q4. Determine which of the statements is incorrect regarding this figure:
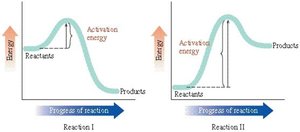
Background
Topic: Reaction Energy Diagrams
This question tests your understanding of energy profiles for chemical reactions, including activation energy, exergonic and endergonic reactions, and how to interpret reaction progress diagrams.
Key Terms and Concepts:
Activation Energy: The minimum energy required for a reaction to occur.
Exergonic Reaction: A reaction where products have lower free energy than reactants (energy is released).
Endergonic Reaction: A reaction where products have higher free energy than reactants (energy is absorbed).
Reaction Rate: Influenced by activation energy; lower activation energy generally means a faster reaction.
Step-by-Step Guidance
Examine the energy diagrams for both reactions. Notice the relative positions of reactants and products for each.
Identify which reaction is exergonic and which is endergonic by comparing the energy levels of reactants and products.
Compare the activation energies for Reaction I and Reaction II by looking at the height of the energy barrier.
Consider how activation energy affects reaction rate and which reaction would occur faster based on the diagrams.
Try solving on your own before revealing the answer!
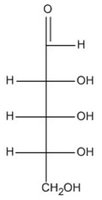
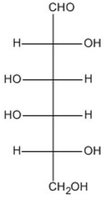
Q15. Which of these molecules is an aldopentose?
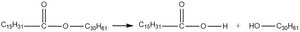
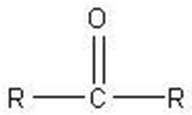
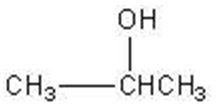
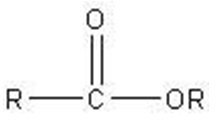
Background
Topic: Carbohydrate Classification
This question tests your ability to identify aldopentoses, which are monosaccharides with five carbon atoms and an aldehyde group.
Key Terms:
Aldopentose: A five-carbon sugar with an aldehyde functional group.
Monosaccharide: The simplest form of carbohydrate.
Functional Group: Aldehyde group is characterized by $-CHO$ at the end of the molecule.
Step-by-Step Guidance
Count the number of carbon atoms in each structure to identify pentoses (five carbons).
Look for the presence of an aldehyde group ($-CHO$) at the end of the molecule.
Distinguish between aldoses (aldehyde group) and ketoses (ketone group).
Compare the structures to see which one fits the definition of an aldopentose.
Try solving on your own before revealing the answer!
Q17. Which of the following is a pyranose ring?
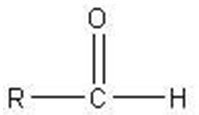
Background
Topic: Carbohydrate Ring Structures
This question tests your ability to distinguish between pyranose and furanose rings in monosaccharides.
Key Terms:
Pyranose: A six-membered ring structure (five carbons and one oxygen).
Furanose: A five-membered ring structure (four carbons and one oxygen).
Monosaccharide: Simple sugar molecule that can cyclize to form rings.
Step-by-Step Guidance
Count the number of atoms in each ring (including oxygen) to determine if it is a pyranose (six-membered) or furanose (five-membered).
Identify the position of the oxygen atom in the ring.
Compare the two structures to see which one matches the pyranose definition.
Recall that pyranose rings are common in glucose and other hexoses.
Try solving on your own before revealing the answer!
Q20. What type of glycosidic bond is shown here?
Background
Topic: Glycosidic Bonds in Disaccharides
This question tests your ability to identify the type of glycosidic linkage between two monosaccharide units.
Key Terms:
Glycosidic Bond: The covalent bond formed between two monosaccharides.
Alpha ($\alpha$) and Beta ($\beta$) Linkages: Refers to the orientation of the bond.
(1→4) Bond: Connects carbon 1 of one sugar to carbon 4 of another.
Step-by-Step Guidance
Identify the carbons involved in the bond (carbon 1 of the first ring and carbon 4 of the second ring).
Determine if the bond is alpha ($\alpha$) or beta ($\beta$) based on the orientation of the oxygen bridge.
Recall the common glycosidic bonds found in disaccharides like maltose, lactose, and sucrose.
Compare the structure to textbook examples to confirm the bond type.
Try solving on your own before revealing the answer!
Q34. The double bonds in this unsaturated triglyceride are:
Background
Topic: Lipid Structure and Unsaturation
This question tests your ability to identify the configuration (cis or trans) of double bonds in fatty acid chains of triglycerides.
Key Terms:
Triglyceride: A lipid molecule made of three fatty acid chains attached to a glycerol backbone.
Unsaturated Fatty Acid: Contains one or more double bonds in the hydrocarbon chain.
Cis and Trans Double Bonds: Refers to the arrangement of hydrogen atoms around the double bond.
Step-by-Step Guidance
Locate the double bonds in the fatty acid chains of the triglyceride structure.
Examine the orientation of the hydrogen atoms around each double bond to determine if they are cis (same side) or trans (opposite sides).
Recall that cis double bonds cause a bend in the chain, while trans double bonds keep the chain straighter.
Compare the structure to typical examples of cis and trans unsaturated fatty acids.
Try solving on your own before revealing the answer!
