 Back
BackExam 1 Review: Chemistry in Our Lives, Measurements, Matter and Energy
Study Guide - Smart Notes

Chapter 1: Chemistry in Our Lives
Chemistry and Chemicals
Chemistry is the scientific study of matter, its properties, and the changes it undergoes. Chemicals are substances with a definite composition, found both naturally and synthetically.
Chemistry: The study of matter and its interactions.
Chemicals: Substances such as water, sodium chloride, and glucose.
Matter: Anything that has mass and occupies space. Examples include air, water, and rocks.
Scientific Method
The scientific method is a systematic approach to investigation, involving observation, hypothesis formation, experimentation, and conclusion.
Observation: Gathering information.
Hypothesis: Proposed explanation.
Experiment: Testing the hypothesis.
Conclusion: Interpreting results.
Math Skills for Chemistry
Mathematical skills are essential for solving chemistry problems, including understanding place values, using positive and negative numbers, calculating percentages, and solving equations.
Place Values: Understanding the value of digits in numbers.
Percentages: Calculating proportions and changes.
Solving Equations: Manipulating algebraic expressions.
Interpreting Graphs: Analyzing data visually.
Scientific and Standard Notation
Scientific notation expresses numbers as a product of a coefficient and a power of ten, useful for very large or small values.
Convert from standard to scientific notation: Move the decimal point and adjust the exponent.
Convert from scientific to standard notation: Reverse the process.
Example:
Chapter 2: Chemistry and Measurements
Units of Measurement
Measurements in chemistry use standardized units for length, mass, time, temperature, and volume. The metric system is preferred for scientific work.
Metric System: Uses units such as meter (m), gram (g), second (s), liter (L), and kelvin (K).
English System: Uses units such as inch, pound, and gallon.
Metric Prefixes and Values
Metric prefixes indicate multiples or fractions of base units.
Mega (M):
Kilo (k):
Deci (d):
Centi (c):
Milli (m):
Micro (\mu):
Temperature Scales
Temperature is measured in Celsius (°C), Fahrenheit (°F), and Kelvin (K). Conversion equations are essential.
Fahrenheit to Celsius:
Celsius to Kelvin:
Measured Numbers and Significant Figures
Significant figures reflect the precision of measured values. Calculations must account for significant figures.
Exact Numbers: Values known with complete certainty.
Rounding: Adjusting numbers to reflect correct precision.
Addition/Subtraction: Result has same decimal places as least precise value.
Multiplication/Division: Result has same significant figures as least precise value.
Prefixes, Equalities, and Conversion Factors
Conversion factors are ratios used to convert between units.
Dimensional Analysis (Factor-Label Method): Systematic approach to unit conversion.
Example:
Density
Density is the mass per unit volume of a substance.
Formula:
Volume of a rectangular solid:
Volume of a cylinder:
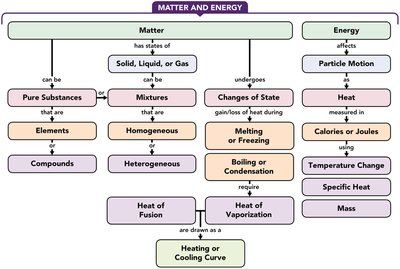
Chapter 3: Matter and Energy
Classification of Matter
Matter can be classified as pure substances or mixtures. Pure substances include elements and compounds, while mixtures can be homogeneous or heterogeneous.
Pure Substances: Have a fixed composition (elements, compounds).
Mixtures: Combinations of two or more substances (homogeneous: uniform, heterogeneous: non-uniform).
States and Properties of Matter
Matter exists in three states: solid, liquid, and gas. Physical properties can be observed without changing the substance, while chemical properties describe the ability to change into another substance.
Physical Change: Alters physical properties, not composition.
Chemical Change: Produces new substances with different properties.
Example: Melting ice (physical), burning wood (chemical).
Temperature Calculations
Temperature conversions are important for scientific measurements.
Celsius to Fahrenheit:
Celsius to Kelvin:
Energy: Potential and Kinetic
Energy is the ability to do work. It exists as potential (stored) or kinetic (motion).
Potential Energy: Stored energy (e.g., chemical bonds).
Kinetic Energy: Energy of motion (e.g., moving particles).
Units: Calorie (cal), kilocalorie (kcal), joule (J), kilojoule (kJ).
Calculating Food Energy
The energy content of food is measured in kilocalories (kcal) or kilojoules (kJ), derived from carbohydrates, fats, and proteins.
Nutritional Calorie: 1 kcal = 1000 cal.
Energy Calculation: Sum of energy from macronutrients.
Specific Heat and Heat Calculations
Specific heat is the energy required to raise the temperature of 1 g of a substance by 1 °C. Heat loss or gain is calculated using mass, specific heat, and temperature change.
Formula:
q: Heat (J or cal)
m: Mass (g)
c: Specific heat (J/g°C or cal/g°C)
ΔT: Change in temperature (°C)
Changes of State and Energy Calculations
Changes of state include melting, freezing, boiling, condensation, and sublimation. Energy is absorbed or released during these processes.
Evaporation: Liquid to gas.
Boiling: Vaporization at boiling point.
Sublimation: Solid to gas.
Heat of Fusion: Energy required to melt 1 g of solid.
Heat of Vaporization: Energy required to vaporize 1 g of liquid.
Heating/Cooling Curve: Graph showing temperature and state changes as heat is added or removed.
Example: Calculating total heat for melting and heating a substance involves summing energy for temperature change and state change.
Additional info: The included concept map visually organizes the relationships between matter, energy, states, changes of state, and measurement units, reinforcing the structure of Chapter 3 topics.
