 Back
BackExam 1 Review Guidance: Chapters 1, 2, 3, 4, 6 (GOB Chemistry)
Study Guide - Smart Notes

Q1. Write scientific notation of 34600000 or 0.000020 with proper SFs.
Background
Topic: Scientific Notation & Significant Figures
This question tests your ability to convert numbers to scientific notation and apply the rules for significant figures (SFs).
Key Terms and Formulas:
Scientific notation: Expresses numbers as where is a number between 1 and 10.
Significant figures: Digits that carry meaning contributing to a measurement's precision.
Step-by-Step Guidance
Identify the number of significant figures in each number (e.g., 34600000 and 0.000020).
Rewrite each number in scientific notation, making sure to preserve the correct number of significant figures.
For 34600000, move the decimal point so only one nonzero digit remains to the left of the decimal.
For 0.000020, do the same, and note the placement of zeros for significant figures.
Try solving on your own before revealing the answer!
Final Answer:
34600000 with proper SFs: (if 3 SFs are intended)
0.000020 with proper SFs: (if 2 SFs are intended)
Scientific notation helps clarify the precision of measurements by showing significant figures explicitly.
Q2. 2.50/3.44 + 539.1 = ? with proper SFs.
Background
Topic: Significant Figures in Calculations
This question tests your ability to apply significant figure rules in division and addition.
Key Terms and Formulas:
Division: The result should have the same number of SFs as the value with the fewest SFs.
Addition: The result should have the same decimal places as the value with the fewest decimal places.
Step-by-Step Guidance
Divide 2.50 by 3.44. Determine the number of SFs in each value.
Round the result of the division to the correct number of SFs.
Add the rounded division result to 539.1.
Round the final sum to the correct decimal place based on the addition rule.
Try solving on your own before revealing the answer!
Final Answer:
2.50/3.44 = 0.726 (rounded to 3 SFs), 0.726 + 539.1 = 539.8 (rounded to tenths place).
Proper SFs ensure the result reflects the precision of the original measurements.
Q3. Be familiar with unit prefixes
Background
Topic: Metric System Prefixes
This question tests your knowledge of metric prefixes, their symbols, values, and how to use them in conversions.
Key Terms and Formulas:
Prefixes: kilo (k), mega (M), giga (G), etc.
Conversion factors: Used to convert between units (e.g., 1 km = m).
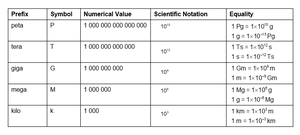
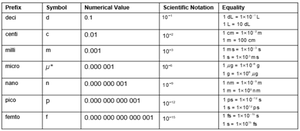
Step-by-Step Guidance
Review the table of prefixes and their corresponding values and symbols.
Practice converting between units using the provided equalities (e.g., meters to kilometers).
Understand how scientific notation relates to each prefix.
Try solving on your own before revealing the answer!
Final Answer:
Knowing prefixes allows you to quickly convert between units and understand scientific notation in chemistry.
Q4. Temperatures: TF = 1.8 TC + 32; TK = TC + 273
Background
Topic: Temperature Conversions
This question tests your ability to convert between Celsius, Fahrenheit, and Kelvin.
Key Terms and Formulas:
(Fahrenheit from Celsius)
(Kelvin from Celsius)
Step-by-Step Guidance
Identify the starting temperature and the scale you need to convert to.
Plug the value into the appropriate formula.
Check your units and make sure you use the correct formula for the conversion.
Try solving on your own before revealing the answer!
Final Answer:
For example, to convert 25°C to Fahrenheit: ; to Kelvin: K.
Temperature conversions are essential for many chemistry calculations.
Q5. Identify the equality and two conversion factors. Calculate using CFs.
Background
Topic: Dimensional Analysis & Conversion Factors
This question tests your ability to identify equalities and use conversion factors to solve unit conversion problems.
Key Terms and Formulas:
Equality: Relationship between two units (e.g., 1 in. = 2.54 cm).
Conversion factor: Fraction used to convert from one unit to another.
Step-by-Step Guidance
Identify the equality (e.g., 1 in. = 2.54 cm).
Write two conversion factors: and .
Set up the calculation using the appropriate conversion factor for the direction you need.
Multiply the given value by the conversion factor, canceling units as needed.
Try solving on your own before revealing the answer!
Final Answer:
For 69440 in. to meters:
Dimensional analysis ensures you use the correct conversion factors and units.
Q6. Density definition? Convert 3.40kg water into mL.
Background
Topic: Density & Unit Conversion
This question tests your understanding of density and your ability to convert mass to volume using density.
Key Terms and Formulas:
Density:
Water density: (at room temperature)
Step-by-Step Guidance
Write the definition of density.
Convert 3.40 kg to grams ().
Use the density of water to set up the conversion: .
Plug in the mass and density values to solve for volume in mL.
Try solving on your own before revealing the answer!
Final Answer:
3.40 kg = 3400 g;
Density allows you to convert between mass and volume for substances.
Q7. How do you define heat as a form of energy? Interconvert units J, kJ, cal, kcal, Cal in food industry.
Background
Topic: Heat & Energy Units
This question tests your understanding of heat as energy and your ability to convert between different energy units.
Key Terms and Formulas:
Heat: Energy transferred due to temperature difference.
Unit conversions: , ,
Step-by-Step Guidance
Define heat as a form of energy.
List the relationships between units (J, kJ, cal, kcal, Cal).
Set up conversion factors for each unit.
Practice converting a value from one unit to another using the conversion factors.
Try solving on your own before revealing the answer!
Final Answer:
Heat is energy transferred between substances due to temperature difference.
Unit conversions are essential for comparing energy values in chemistry and nutrition.
Q8. The energy of a 100g food sample is measured in a calorimeter with 1000.g water. Temperature rises from 25 to 60.°C after burning. How much energy in J is there in the sample? Pay attention to SF.
Background
Topic: Calorimetry & Energy Calculation
This question tests your ability to calculate energy using calorimetry and apply significant figures.
Key Terms and Formulas:
Specific heat ():
Energy ():
For water:
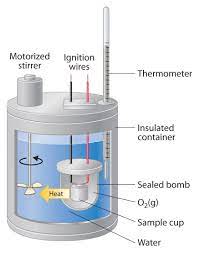
Step-by-Step Guidance
Identify the mass of water, temperature change (), and specific heat.
Calculate .
Plug values into the formula: .
Multiply to find the energy absorbed by the water, which equals the energy released by the food sample.
Try solving on your own before revealing the answer!
Final Answer:
(rounded for SFs)
Calorimetry measures energy content by observing temperature changes in water.
Q9. Be able to find information from periodic table, e.g., symbols, atomic number, atomic mass, valence electrons, ion charges, number of bonds, variable charges, trend in electronegativity, properties such as conductivity, classifications, and etc.
Background
Topic: Periodic Table & Element Properties
This question tests your ability to extract information from the periodic table and understand element properties.
Key Terms and Formulas:
Atomic number: Number of protons
Atomic mass: Weighted average mass of isotopes
Valence electrons: Electrons in the outermost shell
Ion charges: Charge after gaining/losing electrons
Electronegativity: Tendency to attract electrons
Step-by-Step Guidance
Locate the element on the periodic table and identify its symbol, atomic number, and atomic mass.
Determine the number of valence electrons based on its group.
Identify possible ion charges and bonding patterns.
Observe trends in electronegativity and conductivity across periods and groups.
Try solving on your own before revealing the answer!
Final Answer:
Periodic table provides essential information for predicting element behavior and properties.
Q10. Can you tell the atomic number, mass number, # proton, # electrons, # neutrons in oxygen? How many electron levels? Valence electrons? Give an example of its isotope and write the symbol.
Background
Topic: Atomic Structure & Isotopes
This question tests your understanding of atomic structure and isotope notation.
Key Terms and Formulas:
Atomic number (): Number of protons
Mass number ():
Isotope: Same element, different number of neutrons
Symbol:
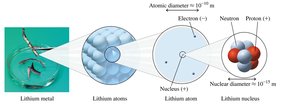
Step-by-Step Guidance
Find oxygen's atomic number (8) and typical mass number (16).
Calculate number of protons, electrons (for neutral atom), and neutrons.
Determine the number of electron levels (period number).
Identify valence electrons (group number).
Write the isotope symbol for oxygen-18.
Try solving on your own before revealing the answer!
Final Answer:
Oxygen: , , 8 protons, 8 electrons, 8 neutrons; isotope:
Isotopes differ in neutron number but have the same chemical properties.
Q11. How many valence electrons in molecule O2?
Background
Topic: Valence Electrons & Molecular Structure
This question tests your ability to count valence electrons in a molecule.
Key Terms and Formulas:
Valence electrons: Electrons in the outermost shell
Oxygen atom: 6 valence electrons
Step-by-Step Guidance
Determine the number of valence electrons for one oxygen atom.
Multiply by two for the O2 molecule.
Check if any electrons are shared or transferred in bonding.
Try solving on your own before revealing the answer!
Final Answer:
O2 molecule has 12 valence electrons (6 per atom).
Valence electrons determine bonding and reactivity.
Q12. What is Rutherford’s atomic model?
Background
Topic: Atomic Models
This question tests your understanding of Rutherford's atomic model and its historical significance.
Key Terms and Formulas:
Rutherford model: Atom has a small, dense, positively charged nucleus; electrons orbit around it.
Gold foil experiment: Showed most alpha particles passed through, some deflected.
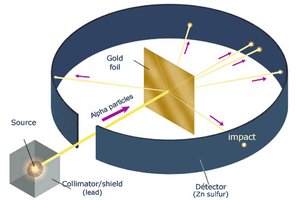
Step-by-Step Guidance
Describe the gold foil experiment and its results.
Explain how the experiment led to the nuclear model of the atom.
Contrast Rutherford's model with previous models (e.g., plum pudding).
Try solving on your own before revealing the answer!
Final Answer:
Rutherford's model: Atom has a dense nucleus with electrons orbiting; most of atom is empty space.
Gold foil experiment provided evidence for the nuclear structure of atoms.
Q13. Name MgCl2? Symbol of Mg cation?
Background
Topic: Naming Ionic Compounds & Ion Symbols
This question tests your ability to name ionic compounds and write ion symbols.
Key Terms and Formulas:
MgCl2: Magnesium chloride
Mg cation:
Step-by-Step Guidance
Identify the cation (Mg) and anion (Cl).
Name the compound using standard rules (metal + nonmetal "-ide").
Write the symbol for the magnesium ion.
Try solving on your own before revealing the answer!
Final Answer:
MgCl2: Magnesium chloride; Mg cation:
Ionic compounds are named by combining cation and anion names.
Q14. Name MnO2? Formula of carbon monoxide, nitrogen dioxide.
Background
Topic: Naming Compounds & Writing Formulas
This question tests your ability to name compounds and write chemical formulas.
Key Terms and Formulas:
MnO2: Manganese(IV) oxide
Carbon monoxide: CO
Nitrogen dioxide: NO2
Step-by-Step Guidance
Identify the oxidation state of manganese in MnO2.
Name the compound using the correct Roman numeral.
Write the formulas for carbon monoxide and nitrogen dioxide.
Try solving on your own before revealing the answer!
Final Answer:
MnO2: Manganese(IV) oxide; CO: carbon monoxide; NO2: nitrogen dioxide.
Correct naming and formula writing are essential for chemical communication.
Q15. Be familiar with common polyatomic ions.
Background
Topic: Polyatomic Ions
This question tests your knowledge of common polyatomic ions and their formulas.
Key Terms and Formulas:
Examples: (nitrate), (sulfate), (ammonium)
Step-by-Step Guidance
Review the names and formulas of common polyatomic ions.
Practice writing their charges and combining them in compounds.
Understand how polyatomic ions affect compound naming.
Try solving on your own before revealing the answer!
Final Answer:
Common polyatomic ions include nitrate (), sulfate (), ammonium ().
Polyatomic ions are groups of atoms with a charge, important in many compounds.
Q16. Draw Lewis structure of NH3
Background
Topic: Lewis Structures
This question tests your ability to draw Lewis structures for molecules.
Key Terms and Formulas:
Lewis structure: Shows valence electrons and bonds
NH3: Ammonia
Step-by-Step Guidance
Count total valence electrons (N: 5, H: 1 each).
Arrange atoms with nitrogen in the center.
Place electrons to form bonds and lone pairs.
Check for octet rule satisfaction.
Try solving on your own before revealing the answer!
Final Answer:
NH3 Lewis structure: N in center, three H atoms bonded, one lone pair on N.
Lewis structures help visualize bonding and electron distribution.
Q17. What is the shape of carbon dioxide?
Background
Topic: Molecular Geometry
This question tests your understanding of molecular shapes based on VSEPR theory.
Key Terms and Formulas:
CO2: Carbon dioxide
Shape: Linear
Step-by-Step Guidance
Draw the Lewis structure for CO2.
Count electron groups around the central atom.
Determine the shape using VSEPR theory.
Try solving on your own before revealing the answer!
Final Answer:
CO2 is linear because there are two electron groups around carbon.
Molecular shape affects physical and chemical properties.
Q18. What does it mean by Trigonal pyramidal, Linear, Bent and Tetrahedral?
Background
Topic: Molecular Geometry & VSEPR Theory
This question tests your understanding of common molecular shapes.
Key Terms and Formulas:
Trigonal pyramidal: 3 bonds, 1 lone pair (e.g., NH3)
Linear: 2 bonds, 0 lone pairs (e.g., CO2)
Bent: 2 bonds, 1 or 2 lone pairs (e.g., H2O)
Tetrahedral: 4 bonds, 0 lone pairs (e.g., CH4)
Step-by-Step Guidance
Review the number of electron groups and lone pairs for each shape.
Visualize the 3D arrangement of atoms for each geometry.
Relate examples to each shape.
Try solving on your own before revealing the answer!
Final Answer:
Each shape is determined by electron groups and lone pairs around the central atom.
VSEPR theory predicts molecular geometry based on repulsion between electron groups.
Q19. How do you define polar or nonpolar bonds?
Background
Topic: Bond Polarity
This question tests your understanding of what makes a bond polar or nonpolar.
Key Terms and Formulas:
Polar bond: Unequal sharing of electrons due to difference in electronegativity
Nonpolar bond: Equal sharing of electrons
Step-by-Step Guidance
Compare electronegativity values of bonded atoms.
If the difference is significant, the bond is polar; if not, it is nonpolar.
Visualize electron distribution in the bond.
Try solving on your own before revealing the answer!
Final Answer:
Polar bonds have unequal electron sharing; nonpolar bonds have equal sharing.
Bond polarity affects molecular properties and interactions.
