 Back
BackExam 4 Practice Problems – Step-by-Step Guidance
Study Guide - Smart Notes

Q3. For the following molecule, which representation correctly represents the bond polarity using delta notation?
Background
Topic: Bond Polarity and Delta Notation
This question tests your understanding of how to represent bond polarity in molecules using the delta notation ( and ), which indicates partial positive and partial negative charges due to differences in electronegativity.
Key Terms:
Bond Polarity: A measure of how electrons are shared between atoms in a bond. If one atom is more electronegative, it attracts electrons more strongly, creating a partial charge.
Delta Notation (, ): Used to show partial positive and negative charges in polar covalent bonds.
Electronegativity: The tendency of an atom to attract electrons in a bond.
Step-by-Step Guidance
Identify the atoms in the molecule and their relative electronegativities. For example, in HF, fluorine is much more electronegative than hydrogen.
Determine which atom will have a partial negative charge () and which will have a partial positive charge (). The more electronegative atom (F) will attract electrons more strongly.
Draw the bond polarity using delta notation: place on the more electronegative atom and on the less electronegative atom.
Check the Lewis structure for lone pairs and the correct placement of charges.
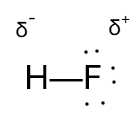
Try solving on your own before revealing the answer!
Final Answer: The correct representation is the image showing H with and F with .
Fluorine is more electronegative, so it attracts electrons more strongly, resulting in a partial negative charge () on F and a partial positive charge () on H.
