 Back
BackExam II Study Guide: Ionic and Molecular Compounds, Chemical Quantities, and Gases
Study Guide - Smart Notes

Chapter 6: Ionic and Molecular Compounds
Definition and Formation of Ionic and Covalent (Molecular) Compounds
Ionic compounds are formed by the transfer of electrons from metals to nonmetals, resulting in the formation of cations (positively charged ions) and anions (negatively charged ions) that are held together by electrostatic forces.
Covalent (molecular) compounds are formed when two or more nonmetals share electrons to achieve a stable electron configuration.
Example: NaCl (ionic), H2O (covalent)
Identification of Ionic and Covalent Compounds
Ionic compounds typically consist of a metal and a nonmetal (e.g., NaCl, MgO).
Covalent compounds are usually composed of nonmetals only (e.g., CO2, NH3).
Naming and Writing Formulas for Ionic Compounds
Write the cation (metal) first, followed by the anion (nonmetal) with its ending changed to “-ide.”
Balance charges to ensure the compound is neutral.
Example: Mg2+ and Cl- combine to form MgCl2.
Monoatomic Cations and Anions
Common cations and anions must be memorized for naming and formula writing. For example, Li+ (lithium), Na+ (sodium), Cl- (chloride), O2- (oxide).
Polyatomic Ions
Polyatomic ions are ions composed of more than one atom, such as NH4+ (ammonium), SO42- (sulfate), and NO3- (nitrate).
Diatomic Elements (Rule of Seven)
Certain elements exist naturally as diatomic molecules: H2, N2, O2, F2, Cl2, Br2, I2.
Octet Rule and Its Exceptions
Atoms tend to gain, lose, or share electrons to achieve eight valence electrons (octet).
Exceptions: Hydrogen seeks a duet (2 electrons), and boron often forms compounds with only 6 electrons.
Electron Dot Formulas (Lewis Structures)
Electron dot formulas represent valence electrons as dots around the element symbol.
Nonmetals share electrons to achieve noble gas configuration.
Typical Bonding Patterns of Nonmetals
The number of bonds formed by nonmetals is related to the number of electrons needed to complete the octet.
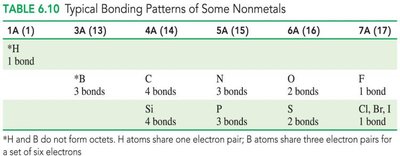
Nomenclature of Molecular Compounds
Prefixes are used to indicate the number of each type of atom in a molecule (e.g., CO2: carbon dioxide).

Electronegativity and Periodic Trends
Electronegativity is the ability of an atom to attract shared electrons in a bond.
Electronegativity increases across a period (left to right) and decreases down a group (top to bottom).
Polarity: Ionic, Polar Covalent, and Nonpolar Covalent Compounds
If the difference in electronegativity between two atoms is large (>1.7), the bond is ionic.
If the difference is moderate (0.4–1.7), the bond is polar covalent.
If the difference is small (<0.4), the bond is nonpolar covalent.
Examples: H2 (nonpolar), HCl (polar), NaCl (ionic)
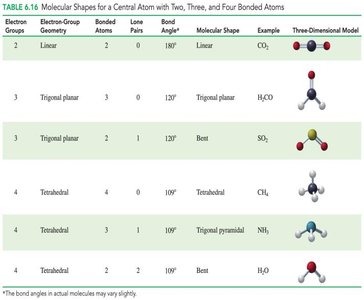
Molecular Shapes, Bond Angles, and Lone Pairs
The shape of a molecule is determined by the number of electron groups, bonded atoms, and lone pairs around the central atom.
Dipole Identification
A dipole exists in a molecule when there is a separation of charge due to differences in electronegativity.
Examples of polar molecules: HCl, NH3, H2O; nonpolar: H2, Cl2, CH4.
Intermolecular Forces
Dipole-dipole interactions: Occur between polar molecules.
Hydrogen bonding: A strong type of dipole-dipole interaction involving H bonded to N, O, or F.
Dispersion forces: Weak forces present in all molecules, especially nonpolar ones.
Chapter 7: Chemical Quantities and Reactions
Definition of Mole and Avogadro’s Number
A mole is the amount of substance that contains particles (Avogadro’s number).
Conversions Involving Moles, Atoms, and Molecules
To convert from moles to atoms/molecules: multiply by Avogadro’s number.
To convert from atoms/molecules to moles: divide by Avogadro’s number.
Example: particles
Moles of Elements in a Formula
The number of moles of each element in a compound is determined by the subscripts in the chemical formula.
Example: 1 mol of H2O contains 2 mol H and 1 mol O.
Molar Mass and Calculations
Molar mass is the mass of one mole of a substance, expressed in grams per mole (g/mol).
To convert between grams and moles:
Balancing Chemical Equations
Ensure the same number of each type of atom on both sides of the equation.
Polyatomic ions that remain unchanged can be balanced as a unit.
Types of Chemical Reactions
Synthesis, decomposition, single replacement, double replacement, and combustion reactions.
Reaction Conditions and Factors Affecting Rate
Temperature, concentration, surface area, and catalysts affect reaction rates.
Exothermic vs. Endothermic Reactions
Exothermic: Energy is released; products have lower energy than reactants.
Endothermic: Energy is absorbed; products have higher energy than reactants.
Chapter 8: Gases
Properties of Gases and Kinetic Theory
Gases have indefinite shape and volume, are compressible, and expand to fill their container.
Kinetic theory: Gas particles are in constant, random motion and collisions are elastic.
Describing Gases: P, V, T, n
P: Pressure
V: Volume
T: Temperature (in Kelvin)
n: Amount of gas (in moles)
Units of Pressure
Pressure can be measured in several units, including atmospheres (atm), millimeters of mercury (mmHg), and torr.

Atmospheric Pressure and Barometers
Atmospheric pressure is the force exerted by air molecules on surfaces; it decreases with altitude and varies with weather.
A barometer is used to measure atmospheric pressure.
Gas Laws
Boyle’s Law: (at constant T and n)
Charles’s Law: (at constant P and n)
Gay-Lussac’s Law: (at constant V and n)
Combined Gas Law:
Avogadro’s Law: (at constant P and T)
STP Conditions and Molar Volume
STP (Standard Temperature and Pressure): 0°C (273 K) and 1 atm.
Molar volume: At STP, 1 mol of any gas occupies 22.4 L.
Dalton’s Law of Partial Pressures
The total pressure of a gas mixture is the sum of the partial pressures of each component gas.
Equation:
Additional info: For all calculations and conceptual questions, refer to worked examples in lecture notes and practice problems for mastery.
