 Back
BackFinal Exam Study Guidance: Molecules, Polarity, and Intermolecular Forces
Study Guide - Smart Notes

Q15. Classify the molecules as polar or nonpolar.
Background
Topic: Molecular Polarity
This question tests your understanding of how to determine whether a molecule is polar or nonpolar based on its structure and the distribution of electrons.
Key Terms:
Polar molecule: A molecule with an uneven distribution of charge, resulting in a dipole moment.
Nonpolar molecule: A molecule with an even distribution of charge, so there is no overall dipole moment.
Electronegativity: The tendency of an atom to attract electrons in a bond.
Lewis structure: A diagram showing the arrangement of atoms and electrons in a molecule.
Step-by-Step Guidance
Examine the Lewis structures provided for each molecule. Identify the atoms and their arrangement.
Determine if there are any differences in electronegativity between the atoms. Large differences can lead to polar bonds.
Assess the molecular geometry. If the shape allows for the dipoles to cancel, the molecule is nonpolar; if not, it is polar.
Look for lone pairs on the central atom, which can affect the shape and polarity.
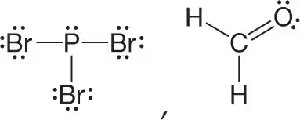
Try solving on your own before revealing the answer!
Final Answer:
Br3P is nonpolar due to its symmetrical arrangement, while H2CO is polar because of its asymmetrical shape and the presence of a highly electronegative oxygen atom.
The polarity of a molecule depends on both the types of atoms present and the overall shape of the molecule.
Q16. Study the branching “R” group of the amino acid serine and choose the single most important type of intermolecular force that the side chain of serine experiences with water.
Background
Topic: Intermolecular Forces
This question tests your ability to identify the dominant intermolecular force between a polar side chain (serine) and water.
Key Terms:
Intermolecular forces: Forces of attraction between molecules.
Hydrogen bond: A strong dipole-dipole interaction between a hydrogen atom bonded to N, O, or F and another electronegative atom.
Dispersion forces: Weak forces present in all molecules, especially nonpolar ones.
Dipole-dipole: Attraction between polar molecules.
Ionic bonds: Attraction between oppositely charged ions.
Step-by-Step Guidance
Examine the structure of serine's side chain and water. Identify the functional groups present.
Determine if there are hydrogen atoms bonded to oxygen or nitrogen, which can participate in hydrogen bonding.
Consider the polarity of the side chain and water. Both are polar, so dipole-dipole and hydrogen bonding are possible.
Evaluate which intermolecular force is strongest and most relevant for interactions between serine's side chain and water.
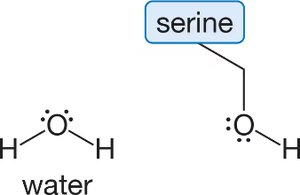
Try solving on your own before revealing the answer!
Final Answer:
The most important intermolecular force between serine's side chain and water is hydrogen bonding.
This is because both serine and water contain O-H groups, which can form strong hydrogen bonds with each other.
