 Back
BackIdentifying Functional Groups and Naming Organic Compounds
Study Guide - Smart Notes

Q26. Which of the following compounds contains a ketone functional group?
Background
Topic: Organic Functional Groups
This question tests your ability to recognize the ketone functional group in organic molecules. Ketones are characterized by a carbonyl group (C=O) bonded to two carbon atoms.
Key Terms and Formulas
Ketone: An organic compound with a carbonyl group (C=O) bonded to two carbon atoms.
Aldehyde: An organic compound with a carbonyl group (C=O) bonded to at least one hydrogen atom.
Alcohol: Contains an -OH (hydroxyl) group.
Ether: Contains an oxygen atom connected to two alkyl or aryl groups (R-O-R').
Step-by-Step Guidance
Examine each structure for the presence of a carbonyl group (C=O).
Determine if the carbonyl group is bonded to two carbon atoms (ketone) or to a hydrogen atom (aldehyde).
Compare the options: Identify which option fits the definition of a ketone.
Eliminate options that are alcohols, ethers, or aldehydes based on their functional groups.
Try solving on your own before revealing the answer!
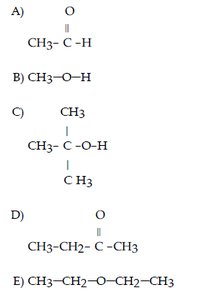
Final Answer: D
Option D contains a carbonyl group (C=O) bonded to two carbon atoms, which is characteristic of a ketone.
The other options represent aldehydes, alcohols, or ethers, not ketones.
Q30. What is the IUPAC name of the following compound?
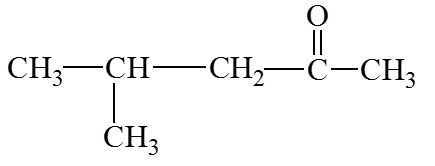
Background
Topic: IUPAC Naming of Ketones
This question tests your ability to apply IUPAC rules to name a ketone with substituents on the carbon chain.
Key Terms and Formulas
IUPAC Naming: Systematic method for naming organic compounds based on their structure.
Ketone: The suffix "-one" is used for ketones.
Substituent: Groups attached to the main carbon chain, such as methyl.
Numbering: Number the chain so the ketone group gets the lowest possible number.
Step-by-Step Guidance
Identify the longest carbon chain containing the ketone group.
Number the chain so the carbonyl carbon gets the lowest possible number.
Identify and name any substituents (e.g., methyl groups) and their positions.
Combine the substituent names and positions with the base name and ketone suffix.
Try solving on your own before revealing the answer!
Final Answer: 4-methyl-2-pentanone
The longest chain is pentane, the ketone is at position 2, and there is a methyl group at position 4.
The correct IUPAC name is 4-methyl-2-pentanone.
