 Back
BackIdentifying the Correct Line Structure for a Branched Alkane
Study Guide - Smart Notes

Q3. Which of the following is a correct line structure for the given molecule?
Background
Topic: Drawing and Interpreting Alkane Structures
This question tests your ability to recognize and match the correct line structure (skeletal formula) for a given branched alkane. Understanding how to interpret and draw line structures is essential for organic chemistry, as it helps visualize the connectivity and branching of carbon atoms in a molecule.
Key Terms and Concepts:
Line Structure (Skeletal Formula): A simplified organic structure where carbon atoms are represented by the ends and intersections of lines, and hydrogen atoms attached to carbons are usually omitted for clarity.
Branched Alkane: An alkane (saturated hydrocarbon) with one or more carbon branches off the main chain.
Longest Chain: The longest continuous chain of carbon atoms in the molecule, which determines the parent name.
Substituents: Groups attached to the main chain, such as methyl or ethyl groups.
Step-by-Step Guidance
Identify the longest continuous chain of carbon atoms in the molecule. This will be the parent chain and determines the base name of the alkane.
Number the carbon atoms in the parent chain from the end nearest a branch (substituent) to give the lowest possible numbers to the substituents.
Locate and identify all branches (alkyl groups) attached to the main chain. Note their positions based on your numbering.
Compare the given line structures to see which one matches the connectivity and branching pattern you have identified.
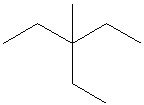
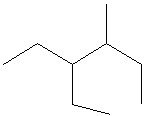
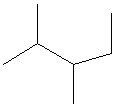
Try solving on your own before revealing the answer!
Final Answer: Structure A is the correct line structure.
Structure A correctly represents the connectivity and branching of the given molecule, matching the positions of all substituents on the parent chain.
