 Back
BackIntermolecular Forces in Ionic and Molecular Compounds
Study Guide - Smart Notes

Chapter 6: Ionic and Molecular Compounds
Introduction to Intermolecular Forces
Intermolecular forces are the attractive forces that exist between molecules and ions in chemical compounds. These forces play a crucial role in determining the physical properties of substances, such as melting points, boiling points, and solubility. Understanding the types and strengths of intermolecular forces is essential for predicting the behavior of ionic and molecular compounds.
Types of Intermolecular Forces
1. Ionic Bonds
Ionic bonds are the electrostatic attractions between positively charged ions (cations) and negatively charged ions (anions). These bonds are typically formed between metals and nonmetals and are the strongest type of attractive force in compounds.
Formation: Transfer of electrons from a metal to a nonmetal.
Example: Sodium chloride (NaCl)
Properties: High melting and boiling points due to strong attractions.
2. Covalent Bonds
Covalent bonds involve the sharing of electrons between nonmetal atoms. While covalent bonds themselves are strong, the forces between covalent molecules (intermolecular forces) can vary in strength.
Formation: Sharing of electrons between nonmetals.
Example: Chlorine gas (Cl2)
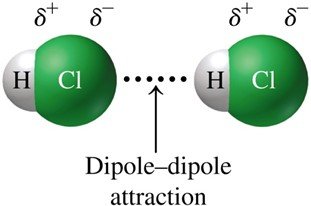
3. Dipole–Dipole Attractions
Dipole–dipole attractions occur between polar molecules, where the positive end of one molecule is attracted to the negative end of another. These forces are stronger than dispersion forces but weaker than hydrogen bonds.
Key Feature: Requires molecules with permanent dipoles (uneven charge distribution).
Example: Hydrogen chloride (HCl)
4. Hydrogen Bonds
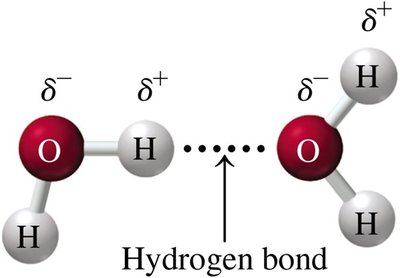
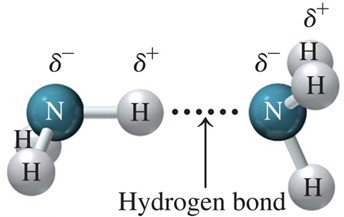
Hydrogen bonds are a special type of dipole–dipole attraction that occurs when hydrogen is bonded to highly electronegative atoms such as fluorine (F), oxygen (O), or nitrogen (N). Hydrogen bonds are the strongest intermolecular force among molecules and are critical in biological structures like DNA.
Key Feature: Hydrogen atom bonded to F, O, or N interacts with a lone pair on another F, O, or N atom.
Examples: Water (H2O), Ammonia (NH3)
5. Dispersion Forces (London Forces)
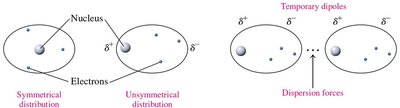
Dispersion forces are weak attractions that occur between nonpolar molecules due to temporary shifts in electron distribution, creating instantaneous dipoles. These forces are present in all molecules but are the only intermolecular force in nonpolar substances.
Key Feature: Temporary dipoles induced by electron movement.
Example: Bromine (Br2), Chlorine (Cl2), Fluorine (F2)
Intermolecular Forces and Melting Points
Relationship Between Force Strength and Melting Point
The strength of intermolecular forces directly affects the melting points of substances. Stronger forces result in higher melting points, while weaker forces lead to lower melting points.
Ionic compounds: Highest melting points due to strong ionic bonds.
Hydrogen bonds: High melting points among molecular compounds.
Dipole–dipole attractions: Moderate melting points.
Dispersion forces: Lowest melting points.
Table: Melting Points of Selected Substances
Type of Force | Substance | Melting Point (°C) |
|---|---|---|
Ionic Bonds | MgF2 | 1248 |
Ionic Bonds | NaCl | 801 |
Hydrogen Bonds | H2O | 0 |
Hydrogen Bonds | NH3 | -78 |
Dipole–Dipole | HI | -51 |
Dipole–Dipole | HBr | -89 |
Dipole–Dipole | HCl | -115 |
Dispersion Forces | Br2 | -7 |
Dispersion Forces | Cl2 | -101 |
Dispersion Forces | F2 | -220 |
Comparison of Bonding and Intermolecular Forces
Strength and Examples
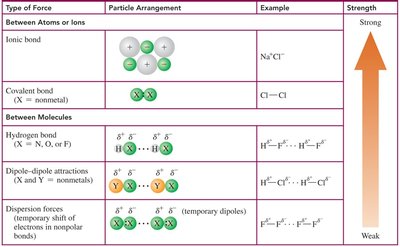
The following table compares the types of bonding and intermolecular forces, their particle arrangements, and examples, ranked from strongest to weakest.
Type of Force | Particle Arrangement | Example | Strength |
|---|---|---|---|
Ionic bond | Alternating positive and negative ions | Na+Cl- | Strong |
Covalent bond | Shared electrons between nonmetals | Cl—Cl | Strong |
Hydrogen bond | H—X (X = N, O, F), dotted line between molecules | H2O, HF | Moderate |
Dipole–dipole attractions | Negative end of one polar molecule attracted to positive end of another | HCl, BrCl | Weak |
Dispersion forces | Temporary dipoles in nonpolar molecules | F2, Cl2 | Weakest |
Identifying Intermolecular Forces in Compounds
Core Chemistry Skill
To identify the main type of intermolecular force present in a compound, consider the nature of the bonding and the elements involved:
Ionic bonds: Present in compounds formed from metals and nonmetals (e.g., KCl).
Hydrogen bonds: Present when H is bonded to F, O, or N (e.g., H2O, NH3).
Dipole–dipole: Present in polar molecules without hydrogen bonding (e.g., NCl3).
Dispersion forces: Present in nonpolar molecules (e.g., Br2).

Concept Map: Ionic and Molecular Compounds
This concept map summarizes the differences between ionic and molecular compounds, their bonding, and the types of intermolecular forces present.
Ionic Compounds: Contain ionic bonds between metals (form positive ions) and nonmetals (form negative ions). Chemical formulas are written using charge balance.
Molecular Compounds: Contain covalent bonds between nonmetals, which can be polar or nonpolar. Molecular compounds are drawn as Lewis structures, with shape determined by VSEPR theory and polarity by electronegativity. Attractive forces include dipole–dipole attractions, hydrogen bonds, or dispersion forces.

Summary Table: Main Types of Intermolecular Forces
Compound | Main Intermolecular Force |
|---|---|
NCl3 | Dipole–dipole forces |
H2O | Hydrogen bonds |
Br2 | Dispersion forces |
KCl | Ionic bonds |
NH3 | Hydrogen bonds |
Key Equations and Concepts
Electronegativity Difference: Determines bond type (ionic, polar covalent, nonpolar covalent).
VSEPR Theory: Predicts molecular shape based on electron pair repulsion.
Charge Balance: Used to write chemical formulas for ionic compounds.
Additional info:
Hydrogen bonds are essential in biological molecules, such as the double helix structure of DNA.
Dispersion forces, though weak, allow nonpolar substances to exist as liquids and solids under certain conditions.
