 Back
BackIntroduction to Chemistry: Matter, Scientific Method, and Scientific Notation
Study Guide - Smart Notes

Introduction to Chemistry
What Is Chemistry?
Chemistry is the scientific study of the composition, structure, properties, and reactions of matter. It is a central science that explains the substances that make up our world and the changes they undergo. Chemistry is present in everyday life, from the air we breathe to the products we use.
Composition: What substances are made of (elements, compounds, mixtures).
Structure: How atoms and molecules are arranged.
Properties: Characteristics such as color, density, melting point, and reactivity.
Reactions: How substances interact and transform into new substances.

Example: An antacid tablet reacts with water, producing bubbles as a result of a chemical reaction.
Matter and Chemicals
Matter refers to anything that has mass and occupies space. Everything around us, including water, air, and living organisms, is made of matter.
Water, oxygen, and carbon dioxide are all examples of matter.
Humans and all living things are composed of matter.
Chemicals are substances with a definite composition and set of properties. They can be naturally occurring or synthesized by chemists. Everyday products, such as toothpaste, are mixtures of various chemicals designed for specific purposes.

Example: Toothpaste contains chemicals for cleaning, whitening, and protecting teeth.
The Scientific Method
Steps of the Scientific Method
The scientific method is a systematic approach used by scientists to explore observations, answer questions, and solve problems. It ensures that scientific inquiry is logical, repeatable, and based on evidence.
Observation: Gather information and ask questions about natural phenomena.
Hypothesis: Propose a tentative explanation or prediction based on observations.
Experiment: Test the hypothesis through controlled experiments.
Conclusion: Analyze results to determine if the hypothesis is supported or refuted.

Example: If you sneeze around cats, you might hypothesize you are allergic. Testing this by visiting homes with and without cats helps confirm or refute your hypothesis.
Application of the Scientific Method in Everyday Life
The scientific method is not limited to laboratories; it can be applied to everyday situations. For example, if you experience allergy symptoms after visiting a friend with a cat, you can use the scientific method to determine the cause.
Observation: Symptoms appear after exposure to a cat.
Hypothesis: You may be allergic to cats.
Experiment: Leave the environment or visit another friend with a cat to see if symptoms recur.
Conclusion: Consistent symptoms support the allergy hypothesis.

Matter and Measurements
Scientific Notation
Scientific notation is a method for expressing very large or very small numbers in a compact form. It is commonly used in chemistry to handle measurements that span many orders of magnitude.
Numbers are written as the product of a coefficient (between 1 and 10) and a power of 10.
Format: where a is the coefficient and n is an integer exponent.
Example: The width of a human hair is meters; the number of hairs on a human scalp is .
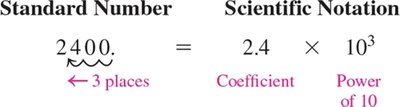
Writing Numbers in Scientific Notation
To convert a standard number to scientific notation:
Move the decimal point to create a coefficient between 1 and 10.
Count the number of places the decimal was moved; this becomes the exponent of 10.
If the decimal is moved to the left, the exponent is positive; if to the right, the exponent is negative.

Examples:
64,000 =
0.021 =
Comparing Standard and Scientific Notation
Quantity | Standard Format | Scientific Notation |
|---|---|---|
Diameter of the Earth | 12,800,000 m | m |
Mass of a human | 68 kg | kg |
Diameter of a chickenpox virus | 0.0000003 cm | cm |
Practice Problems
Write 64,000 in scientific notation:
Write 0.021 in scientific notation:
Select the correct scientific notation for 0.000008:
Select the correct scientific notation for 72,000,000:
Summary: Chemistry is the study of matter and its changes. The scientific method provides a logical framework for investigation. Scientific notation is essential for expressing and working with the wide range of quantities encountered in chemistry.
