 Back
BackIntroduction to Organic Chemistry: Alkanes and Their Properties
Study Guide - Smart Notes

Chapter 12: Introduction to Organic Chemistry
12.1 Organic Compounds
Organic chemistry is the study of compounds containing carbon. The unique bonding properties of carbon allow for a vast diversity of molecular structures, making organic chemistry central to life and industry.

Organic compounds always contain carbon and hydrogen, and may also include other nonmetals such as oxygen, sulfur, nitrogen, phosphorus, or halogens.
Common examples include gasoline, medicines, shampoos, plastics, and perfumes.
Organic compounds typically have low melting and boiling points, are not soluble in water, are less dense than water, and undergo combustion in air.

The element carbon forms the backbone of organic molecules due to its ability to bond with up to four other atoms, creating chains, rings, and complex structures.
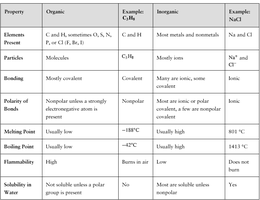
Organic Compounds vs. Inorganic Compounds
Organic and inorganic compounds differ in their elemental composition, bonding, and physical properties.
Property | Organic | Example (C3H8) | Inorganic | Example (NaCl) |
|---|---|---|---|---|
Elements Present | C, H, sometimes O, S, N, P, or halogens | C3H8 | Most metals and nonmetals | Na and Cl |
Particles | Molecules | C3H8 | Mainly ions | Na+ and Cl- |
Bonding | Mostly covalent | Covalent | Many are ionic, some covalent | Ionic |
Polarity of Bonds | Nonpolar unless a strongly electronegative atom is present | Nonpolar | Many are ionic or polar covalent, a few are nonpolar covalent | Ionic |
Melting Point | Usually low | -188°C | Usually high | 801°C |
Boiling Point | Usually low | -42°C | Usually high | 1413°C |
Flammability | High | Burns in air | Does not burn | Does not burn |
Solubility in Water | Not soluble unless a polar group is present | No | Many are soluble unless nonpolar | Yes |
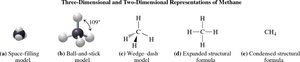
Representation of Carbon Compounds
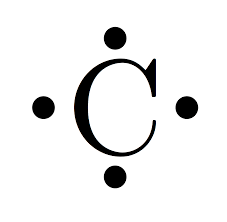
Hydrocarbons are organic compounds consisting only of carbon and hydrogen. Each carbon atom forms four bonds, as seen in methane (CH4), the simplest hydrocarbon.
Methane (CH4): Carbon forms an octet by sharing its four valence electrons with four hydrogen atoms.
12.2 Alkanes
Structure and Naming of Alkanes
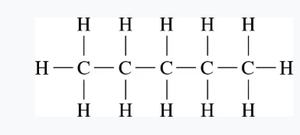
Alkanes are saturated hydrocarbons with only single bonds between carbon atoms. Their names end in "-ane." The IUPAC system is used for systematic naming.
Examples: Methane (1 C), Ethane (2 C), Propane (3 C), Butane (4 C), etc.
For chains with five or more carbons, Greek prefixes are used: pent-, hex-, hept-, oct-, non-, dec-.

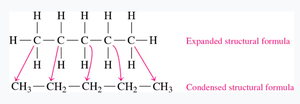
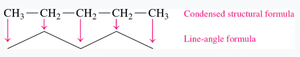
Condensed Structural and Line-Angle Formulas
Organic molecules can be represented in several ways:
Condensed structural formula: Each carbon and its attached hydrogens are grouped together (e.g., CH3CH2CH3).
Line-angle formula: A zigzag line where each vertex and end represents a carbon atom.


Drawing Formulas for Alkanes
Draw the carbon chain.
Add hydrogen atoms to give each carbon four bonds.
Write the condensed structural formula.
Draw the line-angle formula.
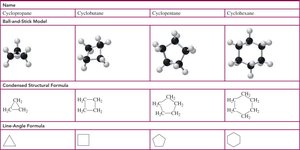
Cycloalkanes
Cycloalkanes are ring-shaped hydrocarbons with two fewer hydrogens than the corresponding straight-chain alkane. They are named by adding the prefix "cyclo-" to the alkane name.
12.3 Alkanes with Substituents
Branched Alkanes and Structural Isomers
When an alkane has four or more carbons, branches (substituents) can form. Compounds with the same molecular formula but different structures are called structural isomers.
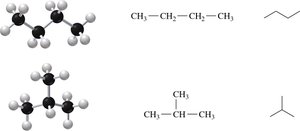
Naming Alkanes with Substituents
Alkyl groups are named by replacing the "-ane" ending with "-yl" (e.g., methyl, ethyl).
Halogen substituents are named as fluoro-, chloro-, bromo-, or iodo-.

Steps for naming:
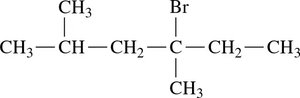
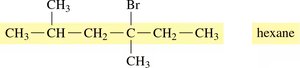
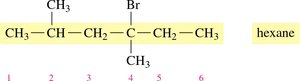
Identify the longest carbon chain (parent alkane).
Number the chain from the end nearest a substituent.
Name and locate each substituent (alphabetically) as a prefix to the main chain.
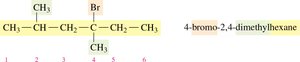
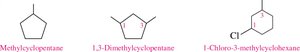
Naming Cycloalkanes with Substituents
For cycloalkanes, if only one substituent is present, no number is needed. If two or more, number the ring to give the lowest numbers to the substituents, starting alphabetically.
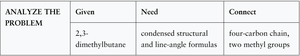
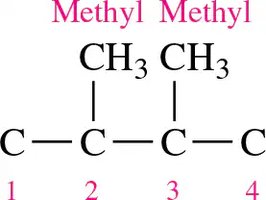
Drawing Structural Formulas for Alkanes with Substituents
To draw condensed and line-angle formulas for branched alkanes:
Draw the main chain.
Number the chain and add substituents at the correct positions.
Add hydrogens to complete four bonds for each carbon.
12.4 Properties of Alkanes
Physical Properties
Alkanes are nonpolar and insoluble in water.
They have densities lower than water (0.62–0.79 g/mL).
They are less dense than water and float on its surface.

Melting and Boiling Points
As the number of carbon atoms increases, melting and boiling points increase due to stronger London dispersion forces.
Branched alkanes have lower boiling points than straight-chain isomers.
Cycloalkanes have higher boiling points than straight-chain alkanes with the same number of carbons.


Chemical Properties: Combustion
Alkanes are chemically stable but undergo combustion in oxygen to produce carbon dioxide, water, and energy.
General equation for alkane combustion:

Additional info:
London dispersion forces are the primary intermolecular forces in alkanes, explaining their low boiling and melting points compared to other organic compounds.
Alkanes are used as fuels (e.g., methane, propane, gasoline) and in products like Vaseline and paraffin wax.
