 Back
BackIntroduction to Organic Chemistry: Hydrocarbons and Nomenclature
Study Guide - Smart Notes

Introduction to Organic Chemistry
Overview of Hydrocarbons
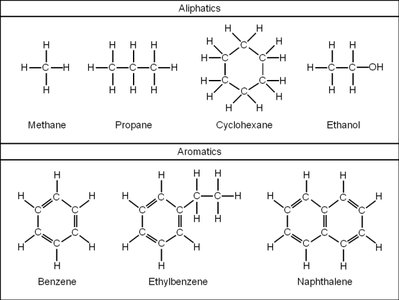
Organic chemistry is the study of compounds primarily composed of carbon and hydrogen. Hydrocarbons are the simplest organic compounds, consisting only of carbon and hydrogen atoms. They are classified into aliphatic (straight or branched chains) and aromatic (ring structures) hydrocarbons.
Aliphatic hydrocarbons: Include alkanes, alkenes, and alkynes.
Aromatic hydrocarbons: Contain benzene rings and related structures.
Examples: Methane, propane, cyclohexane, benzene.
Alkanes: Structure and Nomenclature
Alkanes and Their Properties
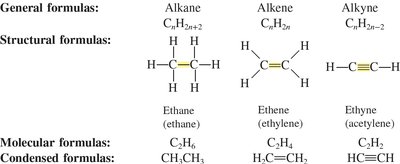
Alkanes are saturated hydrocarbons containing only single bonds between carbon atoms. Their general formula is , and they are non-polar, insoluble in water, but soluble in non-polar solvents. The boiling and melting points increase with molecular weight.
Examples: Methane (), Ethane (), Propane ()
Structural formula: Shows the arrangement of atoms and bonds.
Naming Alkanes (IUPAC System)
The International Union of Pure and Applied Chemistry (IUPAC) system provides rules for naming alkanes:
Step 1: Identify the longest continuous carbon chain (parent hydrocarbon).
Step 2: Number the carbon atoms starting from the end nearest a substituent.
Step 3: Name and locate each substituent (branch), listing them in alphabetical order as prefixes to the main chain name.

Examples of Alkane Nomenclature
Applying the IUPAC rules to various alkane structures:
Example 1: 3-methylheptane (heptane chain with a methyl group at position 3)
Example 2: 4-ethyl-2-methylhexane (hexane chain with ethyl at position 4 and methyl at position 2)
Example 3: 2,3-dimethylpentane (pentane chain with methyl groups at positions 2 and 3)
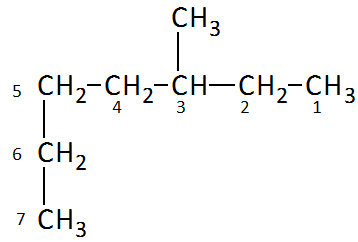
Complex Alkane Nomenclature
When multiple substituents are present, use prefixes (di-, tri-, tetra-) and separate numbers with commas. Branches are listed alphabetically, not by position.
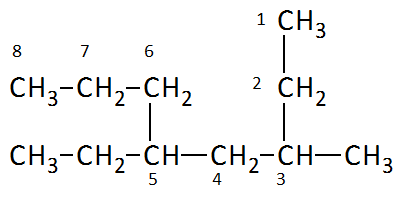
Example: 5-ethyl-3-methyloctane (octane chain with ethyl at position 5 and methyl at position 3)
Example: 3-chloro-2-methylpentane (pentane chain with chloro at position 3 and methyl at position 2)
Haloalkanes
Halogens (F, Cl, Br, I) can replace hydrogen atoms in alkanes. They are named as substituents (fluoro-, chloro-, bromo-, iodo-) and listed alphabetically before the parent chain.
Example: 3-chloro-2-methylpentane
Example: 3,5-difluoro-3-methylheptane
Alkenes and Alkynes: Structure and Nomenclature
Alkenes and Alkynes
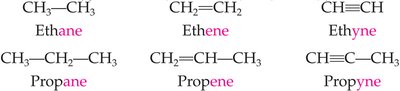
Alkenes contain at least one carbon-carbon double bond (), while alkynes contain at least one carbon-carbon triple bond (). Both are unsaturated hydrocarbons. Their general formulas are for alkenes and for alkynes.
Alkenes: Ethene (), Propene ()
Alkynes: Ethyne (), Propyne ()

Naming Alkenes and Alkynes (IUPAC System)
The IUPAC rules for naming alkenes and alkynes are similar to those for alkanes, with additional steps for double and triple bonds:
Step 1: Name the longest carbon chain containing the double or triple bond.
Step 2: Number the chain from the end nearest the double or triple bond.
Step 3: Indicate the position of the double/triple bond and any substituents.
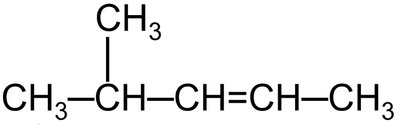
Examples of Alkene and Alkyne Nomenclature
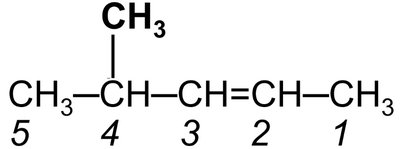
Example 1: 4-methyl-2-pentene (pentene chain with methyl at position 4 and double bond at position 2)
Example 2: 3-methyl-1-butyne (butyne chain with methyl at position 3 and triple bond at position 1)
Example 3: 4-bromo-2-pentyne (pentyne chain with bromo at position 4 and triple bond at position 2)
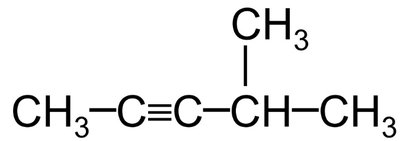
Haloalkenes and Haloalkynes
Halogens can also be substituents in alkenes and alkynes. The double or triple bond takes precedence in numbering, and halogen substituents are listed alphabetically.
Example: 3,5-difluoro-3-methylheptane (heptane chain with fluoro at positions 3 and 5, methyl at position 3)

Summary Table: Hydrocarbon Nomenclature
The following table summarizes the general formulas, structural formulas, and molecular formulas for alkanes, alkenes, and alkynes:
Type | General Formula | Structural Formula | Molecular Formula | Condensed Formula |
|---|---|---|---|---|
Alkane | CnH2n+2 | Single bonds | C2H6 | CH3CH3 |
Alkene | CnH2n | Double bond | C2H4 | H2C=CH2 |
Alkyne | CnH2n-2 | Triple bond | C2H2 | HC≡CH |

Key Points for Exam Preparation
Identify the longest carbon chain for naming.
Number the chain to give substituents the lowest possible numbers.
List substituents alphabetically, using prefixes for multiples.
For alkenes and alkynes, prioritize the double/triple bond in numbering.
Halogen substituents are named as prefixes and listed alphabetically.
