 Back
BackIntroduction to Organic Chemistry: Structure, Properties, and Nomenclature
Study Guide - Smart Notes

Introduction to Organic Chemistry
Definition and Scope
Organic chemistry is the branch of chemistry that studies carbon-based compounds, including biological molecules, drugs, solvents, dyes, and polymers. It does not include most metal salts and many inorganic materials. The field originated from the belief that organic compounds could only be derived from living organisms, but Friedrich Wöhler's synthesis of urea from ammonium cyanate in 1828 disproved this theory.

Organic compounds: Contain carbon and usually hydrogen, sometimes with O, N, S, P, or halogens.
Inorganic compounds: Typically derived from mineral sources and do not primarily contain carbon-hydrogen bonds.
Applications: Organic chemistry is essential for understanding biological processes, pharmaceuticals, and industrial materials.
The Nature of Organic Molecules
Covalent Bonding and Structure
Organic molecules are characterized by covalent bonding, especially between carbon atoms and between carbon and hydrogen. The structure and bonding of carbon atoms are central to organic chemistry.

Carbon forms four covalent bonds, resulting in a tetrahedral geometry with bond angles of 109.5°.
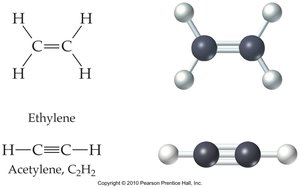
Carbon can form single, double, or triple bonds with other atoms, leading to a variety of molecular structures.
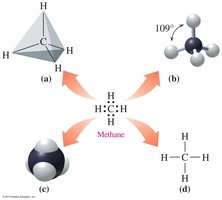
Polarity in Organic Molecules
Organic molecules may contain polar covalent bonds when carbon is bonded to more electronegative elements, such as halogens or oxygen. This results in partial charges and influences molecular interactions.
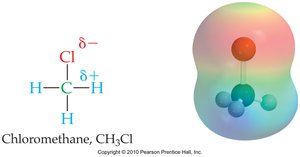
Example: In chloromethane (CH3Cl), chlorine is more electronegative than carbon, creating a polar bond.
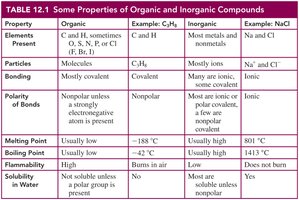
Comparison of Organic and Inorganic Compounds
Organic and inorganic compounds differ in their elemental composition, bonding, physical properties, and solubility.
Property | Organic | Inorganic |
|---|---|---|
Elements Present | C, H, sometimes O, S, N, P, or halogens | Most metals and nonmetals |
Bonding | Mostly covalent | Mostly ionic |
Polarity | Nonpolar or weakly polar | Often ionic or strongly polar |
Melting/Boiling Point | Usually low | Usually high |
Solubility in Water | Low | High |
Families and Functional Groups
Hydrocarbons: Alkanes, Alkenes, Alkynes, and Aromatics
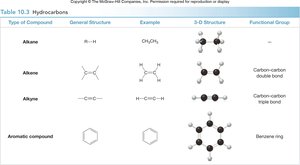
Hydrocarbons are compounds containing only carbon and hydrogen. They are classified based on the types of bonds between carbon atoms:
Alkanes: Only single bonds (saturated hydrocarbons).
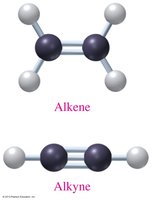
Alkenes: Contain at least one carbon-carbon double bond.
Alkynes: Contain at least one carbon-carbon triple bond.
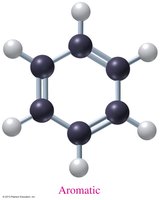
Aromatic compounds: Contain a benzene ring structure.
Type | General Structure | Example | Functional Group |
|---|---|---|---|
Alkane | R–H | CH4 | None |
Alkene | R–CH=CH–R | CH2=CH2 | C=C double bond |
Alkyne | R–C≡C–R | HC≡CH | C≡C triple bond |
Aromatic | C6H6 | Benzene | Benzene ring |
Other Functional Groups
Functional groups are specific groups of atoms within molecules that determine the chemical reactivity and properties of those molecules.
Alcohols: Contain a hydroxyl (–OH) group.
Thiols: Contain a thiol (–SH) group.
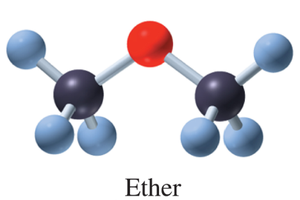
Ethers: Contain an oxygen atom bonded to two carbons (–C–O–C–).
Aldehydes: Contain a carbonyl group (C=O) attached to at least one hydrogen.
Ketones: Contain a carbonyl group attached to two carbons.
Carboxylic acids: Contain a carboxyl group (–COOH).
Esters: Contain a carboxyl group between carbons (–COO–).
Amines: Contain a nitrogen atom.
Amides: Contain a carbonyl group attached to a nitrogen.
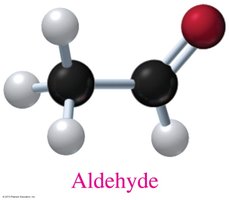
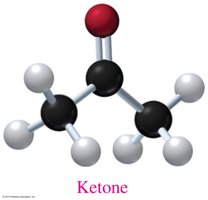


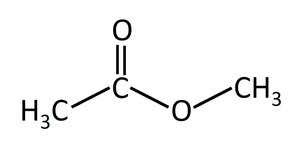
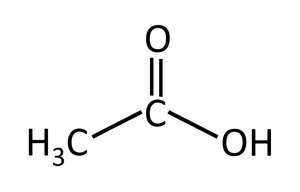
Isomerism in Organic Compounds
Structural and Functional Group Isomers
Isomers are compounds with the same molecular formula but different structural arrangements or functional groups.
Structural isomers: Differ in the connectivity of their atoms.
Functional group isomers: Differ in both molecular connection and family classification.
Naming Organic Compounds (Nomenclature)
Alkanes
Alkanes are named according to the number of carbon atoms in the longest continuous chain (parent chain). Substituents are named and numbered to give the lowest possible numbers to the substituents. The IUPAC system provides systematic rules for naming.
Find the longest carbon chain (parent chain).
Number the chain to give substituents the lowest possible numbers.
Name and number substituents (alkyl groups, halo groups, etc.).
Assemble the name, listing substituents alphabetically and using prefixes (di-, tri-, etc.) for multiples.
Haloalkanes
Halogen atoms (F, Cl, Br, I) can replace hydrogen atoms in alkanes, forming haloalkanes. The halogen is named as a prefix and numbered according to its position on the parent chain.
Example: 2-chloropropane
Physical Properties of Alkanes
Trends and Characteristics
Alkanes are nonpolar, insoluble in water, and have low melting and boiling points that increase with molecular size. Branched alkanes have lower boiling points than their straight-chain isomers due to less efficient packing.
Low-molecular-weight alkanes are gases; medium are liquids; high are waxy solids.
Alkanes are flammable and less dense than water.
Chemical Reactions of Alkanes
Combustion
Alkanes react with oxygen in the presence of a spark or flame to produce carbon dioxide, water, and heat. Incomplete combustion produces carbon monoxide and soot.
General equation:
Halogenation
Halogenation is the replacement of an alkane hydrogen by a halogen atom (Cl or Br), initiated by heat or light. This reaction is used to prepare solvents and other organic molecules.
Cycloalkanes
Structure and Properties
Cycloalkanes are alkanes that contain rings of carbon atoms. They have the general formula CnH2n and exhibit properties similar to acyclic alkanes but with higher boiling points and limited bond rotation.
Examples: Cyclopropane, cyclobutane, cyclohexane
Alkenes, Alkynes, and Aromatic Compounds
Unsaturated Hydrocarbons
Alkenes and alkynes are unsaturated hydrocarbons containing double and triple bonds, respectively. Aromatic compounds contain benzene rings and are characterized by resonance and unique stability.
Naming Alkenes and Alkynes
Longest chain containing the multiple bond is the parent chain.
Number the chain from the end nearest the multiple bond.
Use the suffix –ene for alkenes and –yne for alkynes.
Indicate the position of the multiple bond and substituents.
Cis-Trans Isomerism
Alkenes can exhibit cis-trans (geometric) isomerism due to restricted rotation around the double bond. Cis isomers have substituents on the same side; trans isomers have them on opposite sides.
Addition Reactions of Alkenes
Alkenes undergo addition reactions, such as hydrogenation (addition of H2), hydrohalogenation (addition of HX), halogenation (addition of X2), and hydration (addition of H2O). Markovnikov's rule predicts the major product in unsymmetrical additions.
Example: Hydrogenation
Polymerization
Alkenes can undergo addition polymerization to form polymers, which are large molecules made of repeating monomer units. Examples include polyethylene and polystyrene.
Aromatic Compounds
Benzene and Substituted Benzenes
Benzene (C6H6) is a flat, symmetrical molecule with resonance stabilization. Aromatic compounds are less reactive than alkenes and undergo electrophilic aromatic substitution rather than addition reactions.
Substituted benzenes are named using the parent name benzene, with prefixes for substituents (ortho-, meta-, para- for disubstituted benzenes).
Common names: Toluene (methylbenzene), phenol (hydroxybenzene), aniline (aminobenzene).
Electrophilic Aromatic Substitution
Common reactions include nitration (NO2), halogenation (X), and sulfonation (SO3H) of the benzene ring.
Summary Table: Key Functional Groups
Functional Group | Structure | Example |
|---|---|---|
Alcohol | –OH | Ethanol |
Ether | –O– | Dimethyl ether |
Aldehyde | –CHO | Formaldehyde |
Ketone | –CO– | Acetone |
Carboxylic acid | –COOH | Acetic acid |
Ester | –COO– | Methyl acetate |
Amine | –NH2 | Methylamine |
Amide | –CONH2 | Acetamide |
Additional info: This guide covers the foundational concepts of organic chemistry, including structure, bonding, functional groups, isomerism, nomenclature, and basic reactions. It is suitable for introductory college-level courses and provides a basis for further study in organic and biological chemistry.
