 Back
BackIntroduction to Organic Compounds: Structures, Families, and Isomerism
Study Guide - Smart Notes

Introduction to Organic Compounds
Definition and Classification
Organic compounds are primarily composed of carbon bonded to hydrogen, and may also include oxygen, nitrogen, sulfur, phosphorus, and other elements. They occur naturally as biomolecules (proteins, carbohydrates, lipids, nucleic acids) and can be synthesized in the laboratory.
Inorganic compounds: Do not contain carbon bonded to hydrogen (e.g., CO2, NaHCO3).
Organic compounds: Contain carbon-hydrogen bonds; over 25 million known.
Bonding in Organic Compounds
Covalent bonding is predominant.
Carbon forms four bonds, allowing for chains, branches, and rings.
Other atoms: Nitrogen (3 bonds), Oxygen/Sulfur (2 bonds), Hydrogen/Halogens (1 bond).
Multiple bonds (double, triple) are possible between C, O, N.
Representing Organic Compounds
Types of Formulas
Organic compounds can be represented in several ways, each providing different levels of structural detail.
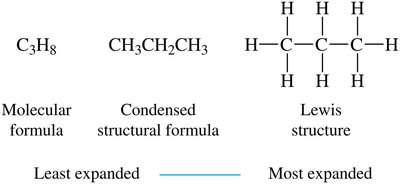
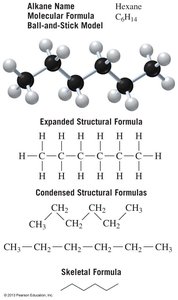
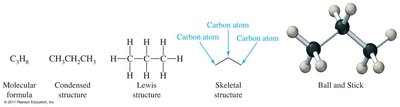
Molecular formula: Shows the number of each type of atom (e.g., C3H8).
Condensed structural formula: Shows each carbon atom and attached hydrogens (e.g., CH3CH2CH3).
Expanded/Lewis structure: Shows every atom and bond.
Skeletal structure: Shows only bonds between carbon atoms; carbon atoms are implied at bond ends and intersections.
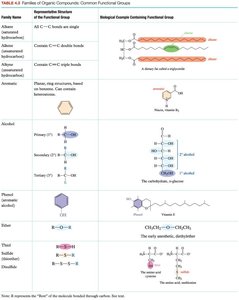
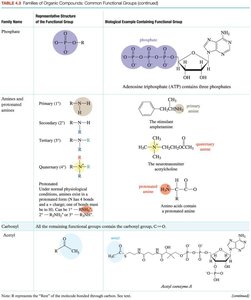
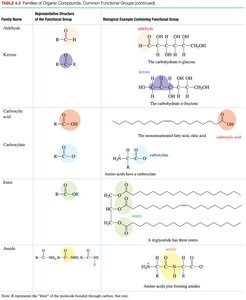
Functional Groups and Families
Functional Groups
Functional groups are specific atoms or groups of atoms within molecules that determine their chemical properties and reactivity.
Represented by 'R' for the rest of the molecule.
Common functional groups include alcohols, ethers, aldehydes, ketones, carboxylic acids, esters, amines, amides, and phosphates.
Members of a family share similar structure and chemical reactions.
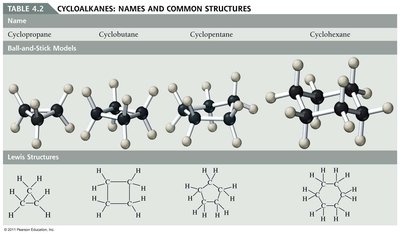
Hydrocarbons: Structure and Classification
Hydrocarbon Families
Hydrocarbons contain only carbon and hydrogen and are classified by the types of bonds between carbon atoms.
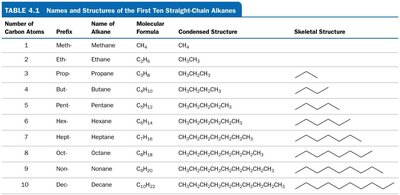
Alkanes: Only single bonds (saturated).
Alkenes: At least one double bond (unsaturated).
Alkynes: At least one triple bond (unsaturated).
Aromatics: Planar ring structures with alternating double and single bonds (e.g., benzene).
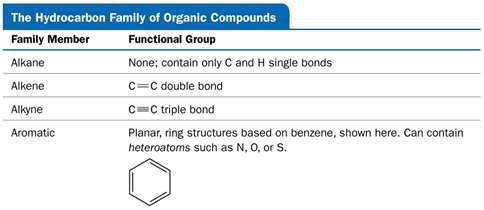

Saturated Hydrocarbons (Alkanes)
Contain only single bonds between carbon atoms.
Can be straight-chain, branched-chain, or cyclic (cycloalkanes).
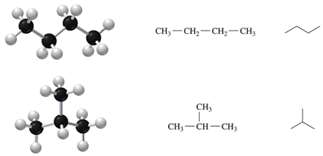
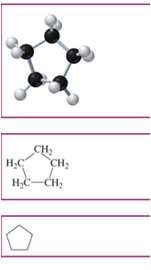
Unsaturated Hydrocarbons
Alkenes: Double bonds; more reactive than alkanes.
Alkynes: Triple bonds; even more reactive, less stable.
Aromatics: Stable rings with resonance; electrons are delocalized.
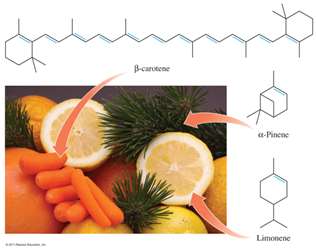

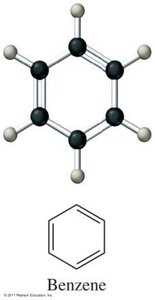
Nomenclature of Organic Compounds
IUPAC Naming System
The International Union of Pure and Applied Chemistry (IUPAC) provides systematic rules for naming organic compounds.
Suffix: Indicates the functional group (e.g., -ane, -ene, -yne).
Prefix: Indicates the number of carbon atoms (e.g., meth-, eth-, prop-, but-).
Branched alkanes: Identify the longest chain (parent), name substituents (alkyl groups), number the chain, and assign locations.
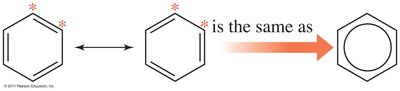
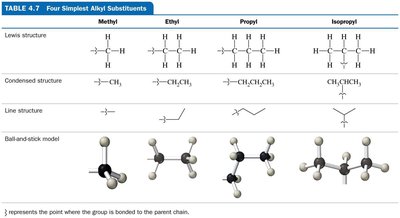
Isomerism in Organic Chemistry
Types of Isomers
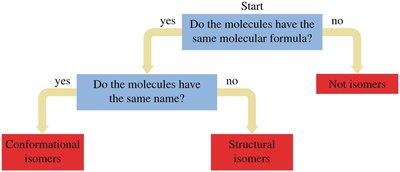
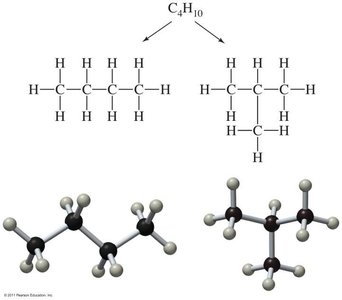
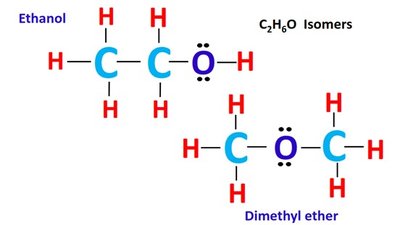
Isomers are compounds with the same molecular formula but different structures.
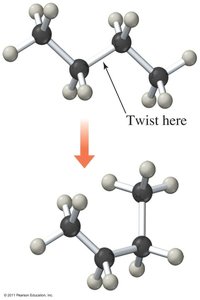
Conformational isomers: Differ by rotation around single bonds; not true isomers.
Structural (constitutional) isomers: Differ in connectivity of atoms.
Stereoisomers: Same connectivity, different spatial arrangement.
Subtypes: Geometric (cis-trans) and enantiomers (mirror images).
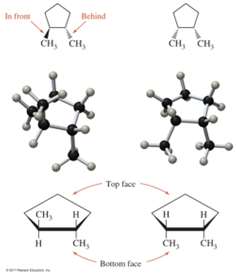
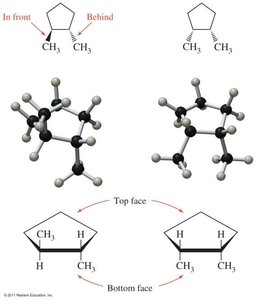
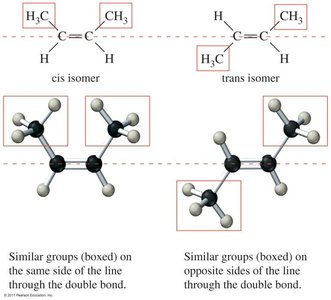
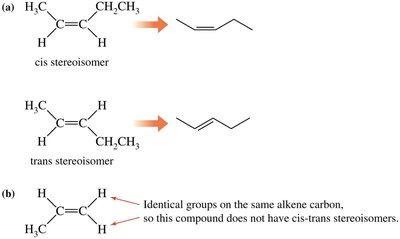
Cis-Trans Isomerism
Occurs in cycloalkanes and alkenes where rotation is restricted.
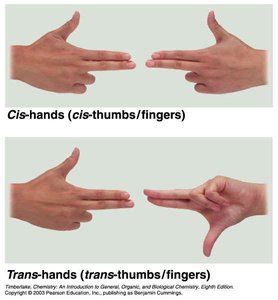
Cis isomer: Groups on the same side.
Trans isomer: Groups on opposite sides.
Enantiomers and Chirality
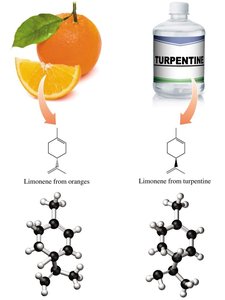
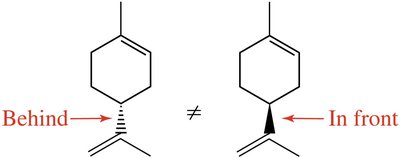
Enantiomers: Non-superimposable mirror images; contain a chiral center (carbon bonded to four different groups).
Chiral objects: Have nonsuperimposable mirror images (e.g., hands, shoes).
Achiral objects: Superimposable on their mirror images (e.g., soccer ball, glass).
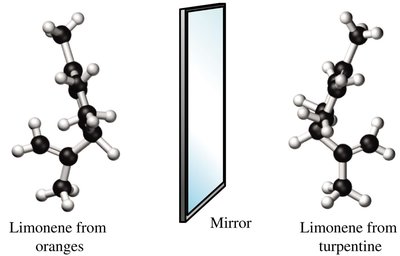


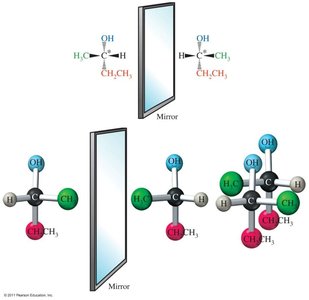
Biological Consequences of Chirality
Chirality in Biology and Pharmaceuticals
Chiral molecules interact differently with biological receptors.
Enantiomers can have different odors, tastes, or biological activity.
Pharmaceuticals: One enantiomer may be beneficial, the other inactive or toxic (e.g., ibuprofen, thalidomide).
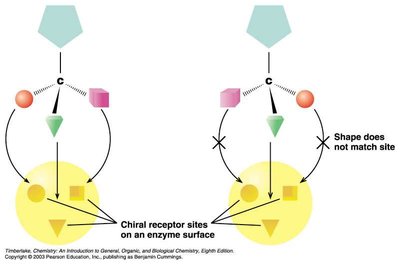
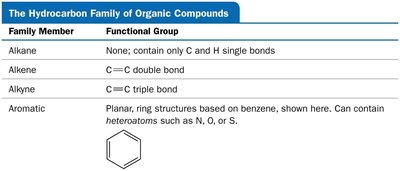
Summary Table: Hydrocarbon Families and Functional Groups
Family Member | Functional Group |
|---|---|
Alkane | None; only C and H single bonds |
Alkene | C=C double bond |
Alkyne | C≡C triple bond |
Aromatic | Planar ring (benzene); may contain heteroatoms |
Summary Table: Common Functional Groups
Family Name | Functional Group | Example |
|---|---|---|
Alcohol | R-OH | Ethanol |
Aldehyde | R-CHO | Formaldehyde |
Ketone | R-CO-R' | Acetone |
Carboxylic Acid | R-COOH | Acetic acid |
Ester | R-COO-R' | Methyl acetate |
Amine | R-NH2 | Methylamine |
Amide | R-CONH2 | Acetamide |
Key Equations and Concepts
General formula for alkanes:
General formula for alkenes:
General formula for alkynes:
Conclusion
Organic chemistry is built upon the understanding of molecular structure, functional groups, and isomerism. Mastery of these concepts is essential for further study in biochemistry, pharmaceuticals, and molecular biology. Additional info: Expanded explanations and tables were added for completeness and clarity.
