 Back
BackIonic and Molecular Compounds: Naming, Formulas, and Polyatomic Ions
Study Guide - Smart Notes

Ch. 6: Ionic and Molecular Compounds
Introduction to Ionic and Molecular Compounds
Ionic and molecular (covalent) compounds are two major classes of chemical compounds, each with distinct bonding and properties. Understanding how to name these compounds and write their chemical formulas is essential in general chemistry.
Types of Compounds
Ionic Compounds: Formed from metals and nonmetals; consist of cations (positive ions) and anions (negative ions).
Molecular (Covalent) Compounds: Formed from nonmetals; atoms share electrons.
Example: Dichlorine monoxide (Cl2O) is a molecular compound because it is composed of two nonmetals.
Naming Ionic Compounds
Ionic compounds are named by stating the cation first, followed by the anion. For compounds containing polyatomic ions, the name of the polyatomic ion is used directly.
Binary Ionic Compounds: Metal + Nonmetal (e.g., CaO: calcium oxide)
With Polyatomic Ions: Use the name of the polyatomic ion (e.g., Mg(NO3)2: magnesium nitrate)
Common Polyatomic Ions
Polyatomic ions are groups of atoms with an overall charge. They can be cations or anions and are commonly found in many compounds.
Ion | Formula | Charge |
|---|---|---|
Ammonium | NH4+ | +1 |
Nitrate | NO3- | -1 |
Sulfate | SO42- | -2 |
Hydroxide | OH- | -1 |
Cyanide | CN- | -1 |
Phosphate | PO43- | -3 |
Carbonate | CO32- | -2 |
Acetate | CH3COO- | -1 |
Writing Formulas for Ionic Compounds
To write the formula for an ionic compound:
Identify the cation and anion (including their charges).
Balance the charges so the net charge is zero.
Write the formula, using parentheses for polyatomic ions if more than one is needed.
Example 1: Magnesium nitrate Cation: Mg2+, Anion: NO3- Balance: Need two NO3- for each Mg2+ Formula: Mg(NO3)2
Example 2: Aluminum hydroxide Cation: Al3+, Anion: OH- Balance: Need three OH- for each Al3+ Formula: Al(OH)3
Example 3: Calcium carbonate Cation: Ca2+, Anion: CO32- Balance: 1:1 ratio Formula: CaCO3
Naming Polyatomic Ionic Compounds
For compounds with polyatomic ions, name the cation first, then the polyatomic anion.
(NH4)2CO3: ammonium carbonate
Li2SO4: lithium sulfate
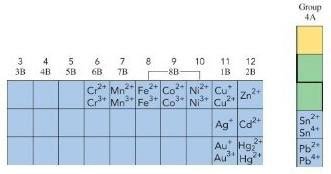
Ionic Compounds with Variable Charge Metals
Some metals (mainly transition metals) can form more than one positive ion. The charge is indicated with Roman numerals in parentheses.
Copper (II) chloride: CuCl2 (Cu2+ + 2Cl-)
Copper (I) chloride: CuCl (Cu+ + Cl-)
Iron (III) fluoride: FeF3 (Fe3+ + 3F-)
Gold (I) oxide: Au2O (2Au+ + O2-)
Writing Formulas from Names
Given the name of an ionic compound, identify the ions and their charges, then balance to write the correct formula.
Sodium sulfate: Na2SO4
Ammonium carbonate: (NH4)2CO3
Magnesium phosphate: Mg3(PO4)2
Naming and Writing Formulas for Molecular Compounds
Molecular compounds are named using prefixes to indicate the number of each atom. The formula is written based on these prefixes.
Mono- (1), Di- (2), Tri- (3), Tetra- (4), Penta- (5), Hexa- (6), etc.
Dihydrogen sulfide: H2S
Dichlorine monoxide: Cl2O
Applications of Ionic Compounds
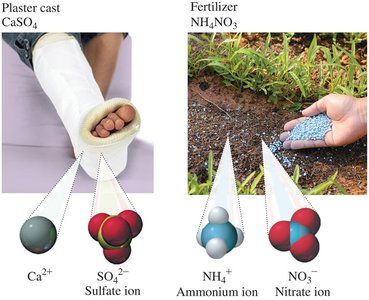
Ionic compounds are widely used in everyday life, such as in medical materials and fertilizers.
Plaster cast: CaSO4 (calcium sulfate) is used for immobilizing broken bones.
Fertilizer: NH4NO3 (ammonium nitrate) provides essential nutrients for plants.
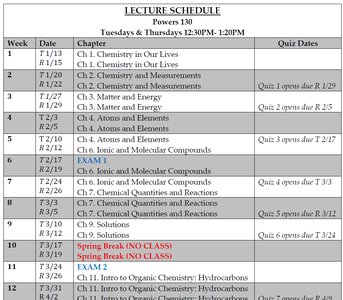
Lecture Schedule and Course Structure
The course covers foundational topics in chemistry, including measurements, matter and energy, atomic structure, chemical bonding, and organic chemistry. Quizzes and exams are scheduled throughout the semester to assess understanding.
Summary Table: Naming and Writing Formulas
Compound Name | Formula | Type |
|---|---|---|
Calcium oxide | CaO | Ionic |
Aluminum sulfide | Al2S3 | Ionic |
Magnesium chloride | MgCl2 | Ionic |
Dichlorine monoxide | Cl2O | Molecular |
Dihydrogen sulfide | H2S | Molecular |
Additional info: The above notes integrate textbook-style explanations, examples, and applications to provide a comprehensive overview of ionic and molecular compounds, as well as their relevance in real-world contexts.
