 Back
BackIonic and Molecular Compounds: Structure, Bonding, and Formulas
Study Guide - Smart Notes

Ions and Ionic Compounds
Formation of Ions
Atoms gain or lose electrons to achieve a stable electron configuration, often resembling the nearest noble gas. The resulting charged species are called ions. Metals typically lose electrons to form cations (positive ions), while nonmetals gain electrons to form anions (negative ions).
Example: Sulfur (S) is in Group 6A and has 6 valence electrons. It gains 2 electrons to form the S2− ion.
Example: Potassium (K) is in Group 1A and loses 1 electron to form K+.
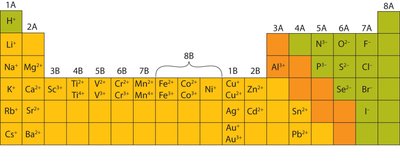
Charges of Transition Metals
Transition metals can form ions with variable positive charges. The charge is indicated by Roman numerals in the name.
Example: Iron can form Fe2+ (iron(II)) and Fe3+ (iron(III)).
Example: Copper can form Cu+ (copper(I)) and Cu2+ (copper(II)).
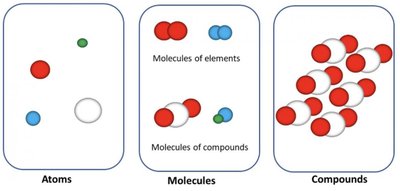
Atoms, Molecules, and Compounds
Definitions and Structure
Understanding the basic building blocks of matter is essential in chemistry:
Atoms: The smallest unit of an element, consisting of protons, neutrons, and electrons.
Molecules: Groups of two or more atoms chemically bonded together. They can be composed of the same or different types of atoms.
Compounds: Substances containing different types of atoms in fixed proportions.
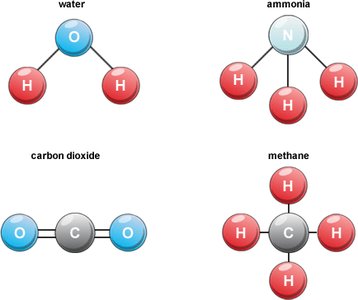
Examples of Molecules and Compounds
Water (H2O): Two hydrogen atoms bonded to one oxygen atom.
Ammonia (NH3): Three hydrogen atoms bonded to one nitrogen atom.
Carbon Dioxide (CO2): Two oxygen atoms bonded to one carbon atom.
Methane (CH4): Four hydrogen atoms bonded to one carbon atom.
Chemical Bonds
Types of Chemical Bonds
Chemical bonds hold atoms together in molecules and compounds. The type of bond depends on the atoms involved:
Ionic Bonds: Formed by the transfer of electrons from a metal to a nonmetal, resulting in the formation of cations and anions.
Covalent Bonds: Formed by the sharing of electrons between nonmetal atoms.

Octet Rule
The octet rule states that atoms tend to gain, lose, or share electrons to achieve eight electrons in their valence shell, resulting in greater stability.
Chemical Formulas
Writing Chemical Formulas
A chemical formula is a combination of element symbols that shows the composition of a compound. Subscripts indicate the number of each atom present.
Example: H2O contains 2 hydrogen atoms and 1 oxygen atom.
Example: Na2SO4 contains 2 sodium atoms, 1 sulfur atom, and 4 oxygen atoms.
Example: Ba(OH)2 contains 1 barium atom, 2 oxygen atoms, and 2 hydrogen atoms.
Counting Atoms in a Formula
Example: Li2SO4 contains 2 lithium atoms, 1 sulfur atom, and 4 oxygen atoms, for a total of 7 atoms.
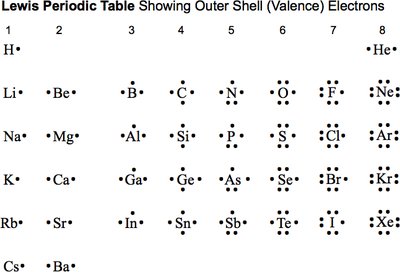
Lewis Symbols and Valence Electrons
Lewis Symbols
Lewis symbols visually represent the valence electrons of an atom. The number of valence electrons corresponds to the group number in the periodic table.
Valence Electrons: Electrons in the outermost shell that participate in chemical bonding.
Example: Nitrogen (N) is in Group 5A and has 5 valence electrons.
Formation and Naming of Ionic Compounds
Steps for Writing Ionic Compound Formulas
Determine the charge of each ion.
Balance the charges so the total positive and negative charges are equal.
Ensure the overall charge of the compound is zero.
List the number of atoms, with the metal first and the nonmetal second.
Example: Magnesium (Mg) forms Mg2+, and chlorine (Cl) forms Cl−. The formula is MgCl2.
Example: Sodium (Na) forms Na+, and sulfur (S) forms S2−. The formula is Na2S.
Example: Calcium (Ca) forms Ca2+, and nitrogen (N) forms N3−. The formula is Ca3N2.
Example: Aluminum (Al) forms Al3+, and oxygen (O) forms O2−. The formula is Al2O3.
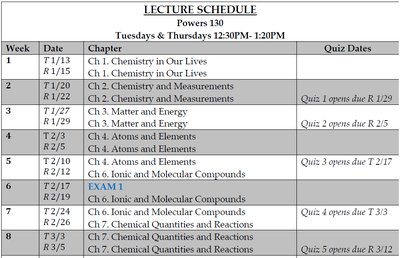
Lecture Schedule and Course Structure
Relevant Chapters and Timeline
The course covers foundational chemistry topics, including ionic and molecular compounds, chemical quantities, and reactions. The schedule outlines the sequence of chapters and quiz dates.
Summary Table: Common Ion Charges
Periodic Table of Ion Charges
The periodic table can be used to predict the common charges of ions formed by elements in each group.

Summary Table: Lewis Symbols
Lewis Periodic Table
This table shows the number of valence electrons for each element, represented as dots around the element symbol.

Visual Representation of Atoms, Molecules, and Compounds
Diagram of Basic Chemical Structures
This diagram visually distinguishes between single atoms, molecules of elements, molecules of compounds, and compounds.

Visual Representation of Covalent Compounds
Structural Diagrams
These diagrams show the arrangement of atoms in common covalent compounds, illustrating the concept of shared electrons.

Key Equations
Formation of Ionic Compounds
General Equation:
Example:
Counting Atoms in a Formula
Example: contains atoms.
Additional info:
Polyatomic ions and Lewis structures for molecules are also part of Chapter 6, but not fully covered in the provided materials.
