 Back
BackIonic and Molecular Compounds: Structure, Bonding, and Formulas
Study Guide - Smart Notes

Atoms, Molecules, and Compounds
Definitions and Classifications
Understanding the basic units of matter is essential in chemistry. Atoms, molecules, and compounds are foundational concepts that describe how elements exist and interact.
Atom: The smallest unit of an element, consisting of protons, neutrons, and electrons. Examples: H, Mg, N.
Molecule: A group of two or more atoms chemically bonded together. Molecules can consist of the same or different types of atoms. Examples: H2 (element), H2O (compound), CO2 (compound).
Compound: A substance containing two or more different types of atoms in fixed proportions. Examples: H2O, CO2, NaCl.
Example: Water (H2O) is a compound made of two hydrogen atoms and one oxygen atom bonded together.
Chemical Bonds
Types of Chemical Bonds
Chemical bonds hold atoms together in molecules and compounds. The type of bond formed depends on the atoms involved and their tendency to gain, lose, or share electrons.
Ionic Bonds: Formed by the transfer of electrons from a metal (cation) to a nonmetal (anion). Example: NaCl (sodium chloride).
Covalent Bonds: Formed by the sharing of electrons between nonmetal atoms. Example: H2O (water), CO2 (carbon dioxide).
Octet Rule: Atoms tend to gain, lose, or share electrons to achieve a stable configuration of eight valence electrons (a 'stable octet').
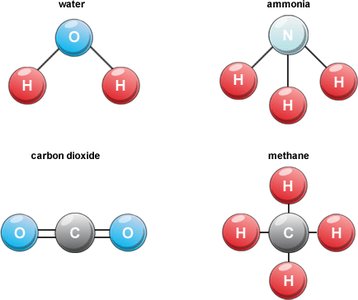
Example: In water (H2O), oxygen shares electrons with two hydrogen atoms, forming covalent bonds.
Ions and Ionic Compounds
Formation of Ions
Atoms form ions by gaining or losing electrons to achieve a stable electron configuration. The charge of an ion depends on the number of electrons lost or gained.
Cation: A positively charged ion formed when an atom loses electrons (usually metals).
Anion: A negatively charged ion formed when an atom gains electrons (usually nonmetals).
Example: Sulfur (S) is in Group 6, has 6 valence electrons, and gains 2 electrons to become S2−.
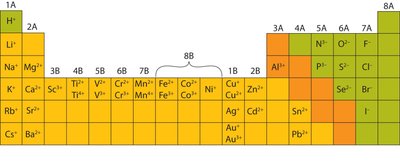
Transition Metals: These elements can form ions with variable positive charges. The charge is indicated with Roman numerals (e.g., Fe(II) is Fe2+, Fe(III) is Fe3+).
Writing Formulas for Ionic Compounds
To write the formula for an ionic compound:
Determine the charge of each ion.
Balance the charges so the total positive and negative charges are equal.
Write the formula with the metal (cation) first, followed by the nonmetal (anion).
Example: Magnesium (Mg) forms Mg2+; chlorine (Cl) forms Cl−. The formula is MgCl2.
Practice: Predict the formulas for the following pairs:
Lithium and oxygen: Li+ and O2− combine to form Li2O.
Aluminum and sulfur: Al3+ and S2− combine to form Al2S3.
Chemical Formulas and Counting Atoms
Interpreting Chemical Formulas
A chemical formula uses element symbols and subscripts to show the types and numbers of atoms in a compound.
H2O: 2 hydrogen atoms, 1 oxygen atom
Na2SO4: 2 sodium atoms, 1 sulfur atom, 4 oxygen atoms
Ba(OH)2: 1 barium atom, 2 oxygen atoms, 2 hydrogen atoms
Example: How many atoms are in Li2SO4? 2 (Li) + 1 (S) + 4 (O) = 7 atoms total.
Lewis Symbols and Valence Electrons
Lewis Symbols
Lewis symbols are a visual shorthand to represent the valence electrons of an atom. The number of valence electrons corresponds to the group number for main group elements.
Dots are placed around the element symbol to represent valence electrons.
These electrons are involved in chemical bonding.
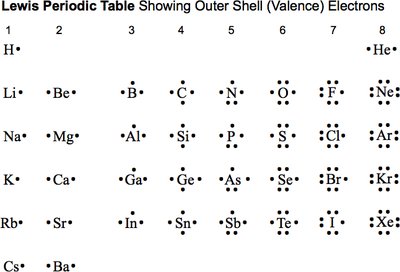
Example: Nitrogen (N) is in Group 5 and has 5 valence electrons, so its Lewis symbol is N with five dots around it.
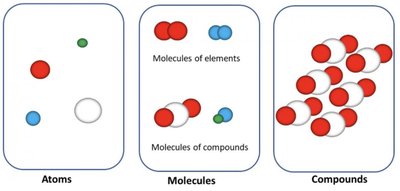
Visualizing Atoms, Molecules, and Compounds
Particle Diagrams
Diagrams can help visualize the differences between atoms, molecules, and compounds:
Atoms: Single spheres representing individual atoms.
Molecules: Groups of spheres joined together, representing bonded atoms. Can be elements (same type) or compounds (different types).
Compounds: Repeating units of molecules with different types of atoms.

Course Structure and Relevance
Lecture Schedule Overview
The course covers foundational topics in chemistry, including:
Chemistry in Our Lives
Chemistry and Measurements
Matter and Energy
Atoms and Elements
Ionic and Molecular Compounds
Chemical Quantities and Reactions
Note: Regular quizzes and exams are scheduled to reinforce learning and assess understanding of these topics.
Summary Table: Common Ion Charges
Group | Common Charge | Example Ion |
|---|---|---|
1A | +1 | Na+ |
2A | +2 | Mg2+ |
3A | +3 | Al3+ |
5A | -3 | N3− |
6A | -2 | O2− |
7A | -1 | Cl− |
Additional info: Transition metals can have variable charges, indicated by Roman numerals in their names (e.g., Fe(II), Fe(III)).
