 Back
BackIonic and Molecular Compounds: Structure, Naming, and Examples
Study Guide - Smart Notes

Ch.6 Ionic and Molecular Compounds
Introduction to Chemical Bonds
Chemical bonds are the forces that hold atoms together in compounds. The two main types of chemical bonds are ionic bonds and covalent bonds, each with distinct characteristics and formation processes.
Ionic bonds: Formed by the transfer of electrons from one atom to another, typically between a metal and a non-metal.
Covalent bonds: Formed by the sharing of electrons between atoms, usually between two non-metals.
Ions and Ionic Compounds
Ions are atoms or groups of atoms that have gained or lost electrons, resulting in a net charge. Ionic compounds are formed when positive ions (cations) and negative ions (anions) combine to balance their charges.
Cation: A positively charged ion (e.g., Mg2+, Na+).
Anion: A negatively charged ion (e.g., Cl-, S2-).
Charge balance: The total positive charge must equal the total negative charge in the compound.
Predicting Ionic Compound Formulas
To predict the formula of an ionic compound, determine the charges of the ions and combine them in ratios that balance the charges.
Example 1: Magnesium and chlorine combine to form MgCl2 ().
Example 2: Magnesium and phosphorus combine to form Mg3P2 ().
Example 3: Lithium and sulfur combine to form Li2S (}).
Table: Ionic Compound Formation Examples
Elements | Cations/Anions | Formula |
|---|---|---|
B and F | B3+, F- | BF3 |
K and S | K+, S2- | K2S |
Mg and N | Mg2+, N3- | Mg3N2 |
Naming Ionic Compounds
The naming of ionic compounds follows a systematic approach:
Identify the cation and anion.
Name the cation using the element's name.
Name the anion by deleting the ending and adding the suffix -ide.
Example: Na2S is named sodium sulfide.
Example: MgO is named magnesium oxide.
Molecular (Covalent) Compounds
Molecular compounds are formed by covalent bonds, which involve the sharing of electrons between two non-metal atoms. Each covalent bond is represented by a single line in structural formulas.
Diatomic elements: H2, N2, O2, F2, Cl2
Common molecules: NH3 (ammonia), CO2 (carbon dioxide), H2O (water), CH4 (methane)
Lewis Structures for Molecules
Lewis structures visually represent the arrangement of electrons in molecules, showing shared pairs (covalent bonds) and lone pairs.
Naming Covalent Compounds
The naming of covalent compounds uses prefixes to indicate the number of each type of atom:
Name the first element using its full name.
Name the second element with the suffix -ide.
Add prefixes to indicate the number of atoms (mono-, di-, tri-, tetra-, etc.). No prefix is used for the first element if it is mono.
Example: CO2 is named carbon dioxide.
Example: S2Br2 is named disulfur dibromide.
Example: N2O5 is named dinitrogen pentoxide.
Table: Prefixes for Covalent Compounds
Number | Prefix |
|---|---|
1 | mono- |
2 | di- |
3 | tri- |
4 | tetra- |
5 | penta- |
6 | hexa- |
7 | hepta- |
8 | octa- |
Common Names of Covalent Compounds
Some covalent compounds have common names that differ from their systematic molecular names. The following table compares these names:
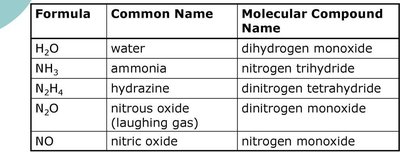
Formula | Common Name | Molecular Compound Name |
|---|---|---|
H2O | water | dihydrogen monoxide |
NH3 | ammonia | nitrogen trihydride |
N2H4 | hydrazine | dinitrogen tetrahydride |
N2O | nitrous oxide (laughing gas) | dinitrogen monoxide |
NO | nitric oxide | nitrogen monoxide |
Lecture Schedule Context
The lecture schedule confirms the relevance of these topics to the course, with Ch.6 "Ionic and Molecular Compounds" covered in weeks 6 and 7. This aligns with the study notes provided above.
 Additional info: The schedule also shows the progression from basic chemistry concepts to more advanced topics, reinforcing the importance of understanding ionic and molecular compounds for later chapters.
Additional info: The schedule also shows the progression from basic chemistry concepts to more advanced topics, reinforcing the importance of understanding ionic and molecular compounds for later chapters.
