 Back
BackIonic and Molecular Compounds: Structure, Properties, and Naming
Study Guide - Smart Notes

Chapter 6: Ionic & Molecular Compounds
Metals vs Nonmetals
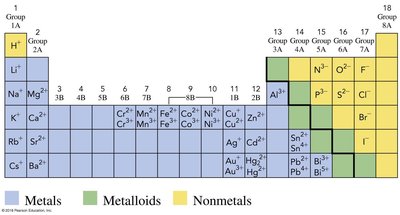
The periodic table is divided into metals, metalloids, and nonmetals, each with distinct chemical behaviors. Metals tend to lose electrons and form cations, while nonmetals gain electrons to form anions. The interaction between these types of elements forms the basis for ionic and molecular compounds.
Metals: Located on the left and center of the periodic table; typically lose electrons.
Nonmetals: Located on the right; typically gain electrons.
Metalloids: Elements with properties intermediate between metals and nonmetals.
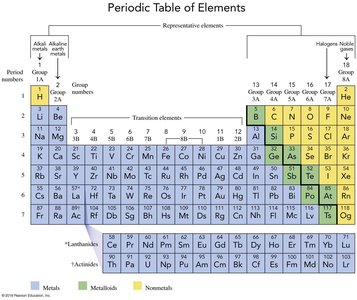
Ionic vs Covalent Bonding
Compounds are formed by either transferring or sharing electrons. Ionic bonds occur between metals and nonmetals, while covalent bonds occur between nonmetals.
Ionic Bond: Formed by transfer of electrons from a metal to a nonmetal, resulting in cations and anions.
Covalent Bond: Formed by sharing electrons between nonmetals.
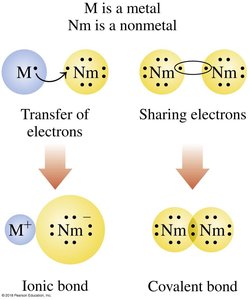
Diatomic Molecular Compounds
Certain elements exist naturally as diatomic molecules, meaning two atoms of the same element are bonded together. These molecules have an oxidation state of zero.
Diatomic Elements: H2, N2, O2, F2, Cl2, Br2, I2
Oxidation State: For diatomic molecules, the charge is zero.
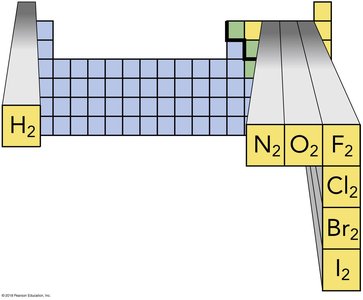
Diatomic Molecule | Name |
|---|---|
H2 | Hydrogen |
N2 | Nitrogen |
O2 | Oxygen |
F2 | Fluorine |
Cl2 | Chlorine |
Br2 | Bromine |
I2 | Iodine |
Formation of Ionic and Molecular Compounds
Ionic compounds are composed of ions, while molecular compounds are composed of molecules. The type of bond and the nature of the particles determine their properties.
Ionic Compounds: Made of ions, held together by ionic bonds (e.g., NaCl).
Molecular Compounds: Made of molecules, held together by covalent bonds (e.g., H2O, C2H6).
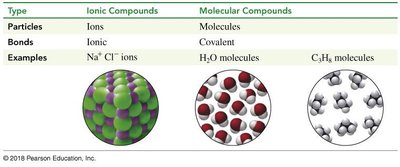
Type | Ionic Compounds | Molecular Compounds |
|---|---|---|
Particles | Ions | Molecules |
Bonds | Ionic | Covalent |
Examples | Na+ Cl- ions | H2O, C2H6 molecules |
Cations and Anions
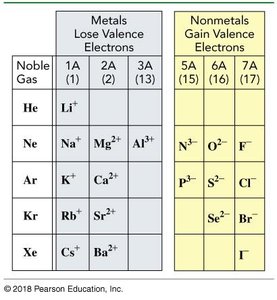
Atoms form ions by losing or gaining electrons. Metals form cations (positive), and nonmetals form anions (negative).
Cation: Positively charged ion formed by loss of electrons (e.g., Na+).
Anion: Negatively charged ion formed by gain of electrons (e.g., Cl-).
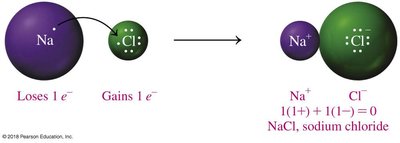
Formation of Sodium and Chloride Ions
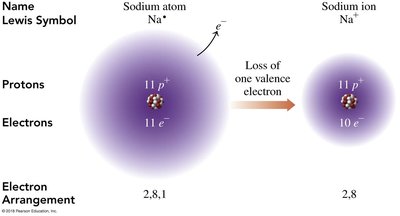
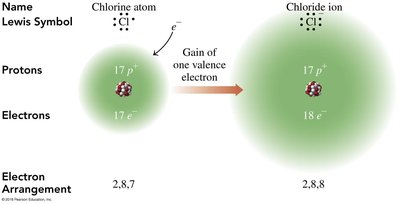
Sodium (Na) loses one electron to become Na+, while chlorine (Cl) gains one electron to become Cl-. This transfer results in the formation of sodium chloride (NaCl).
Electron Arrangement: Sodium changes from 2,8,1 to 2,8; Chlorine changes from 2,8,7 to 2,8,8.
Charge Balance: The sum of charges in NaCl is zero.
Naming Ionic Compounds
Ionic compounds are named by combining the names of the cation and anion. For main group elements, the charge is determined by the group number. Transition metals may have variable charges, indicated by Roman numerals.
Main Group Elements: Charge is consistent within a group.
Transition Metals: Charge varies; oxidation state must be specified.
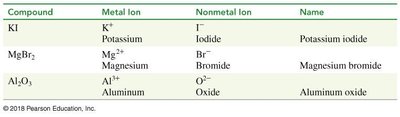
Compound | Metal Ion | Nonmetal Ion | Name |
|---|---|---|---|
KI | K+ | I- | Potassium iodide |
MgBr2 | Mg2+ | Br- | Magnesium bromide |
Al2O3 | Al3+ | O2- | Aluminum oxide |
Variable Charge (Transition Metals)
Transition metals and some other elements (Sn, Pb, Bi) can form ions with different charges. The oxidation state is specified in the compound name.
Element | Possible Ions | Name of Ion |
|---|---|---|
Fe | Fe2+, Fe3+ | Iron(II), Iron(III) |
Cu | Cu+, Cu2+ | Copper(I), Copper(II) |
Sn | Sn2+, Sn4+ | Tin(II), Tin(IV) |
Pb | Pb2+, Pb4+ | Lead(II), Lead(IV) |
Compound | Systematic Name |
|---|---|
FeCl2 | Iron(II) chloride |
Fe2O3 | Iron(III) oxide |
Cu3P | Copper(I) phosphide |
CrBr2 | Chromium(II) bromide |
SnCl2 | Tin(II) chloride |
PbS2 | Lead(IV) sulfide |
BiF3 | Bismuth(III) fluoride |
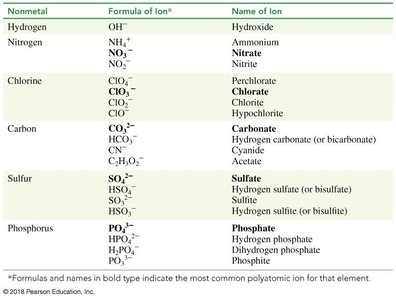
Common Polyatomic Ions
Polyatomic ions are groups of atoms bonded together with an overall charge. They are important in both biological and chemical contexts.
Examples: Hydroxide (OH-), Ammonium (NH4+), Nitrate (NO3-), Carbonate (CO32-), Bicarbonate (HCO3-), Sulfate (SO42-), Phosphate (PO43-).
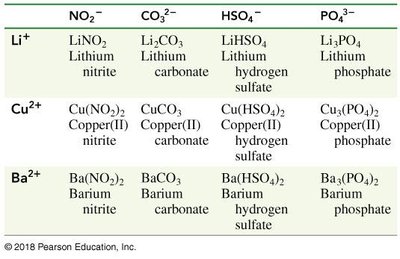
Nonmetal | Formula of Ion | Name of Ion |
|---|---|---|
Hydrogen | OH- | Hydroxide |
Nitrogen | NH4+, NO3-, NO2- | Ammonium, Nitrate, Nitrite |
Chlorine | ClO4-, ClO3-, ClO2-, ClO- | Perchlorate, Chlorate, Chlorite, Hypochlorite |
Carbon | CO32-, HCO3-, CN-, C2H3O2- | Carbonate, Hydrogen carbonate, Cyanide, Acetate |
Sulfur | SO42-, HSO4-, SO32-, HSO3- | Sulfate, Hydrogen sulfate, Sulfite, Hydrogen sulfite |
Phosphorus | PO43-, HPO42-, H2PO4-, PO33- | Phosphate, Hydrogen phosphate, Dihydrogen phosphate, Phosphite |
Common Polyatomic Compounds and Their Uses
Many polyatomic compounds have important medical and industrial uses.
Formula | Name | Medical Use |
|---|---|---|
AlPO4 | Aluminum phosphate | Antacid |
Al2(SO4)3 | Aluminum sulfate | Antiperspirant, anti-infective |
BaSO4 | Barium sulfate | Contrast medium for X-rays |
CaCO3 | Calcium carbonate | Antacid, calcium supplement |
Ca3(PO4)2 | Calcium phosphate | Calcium dietary supplement |
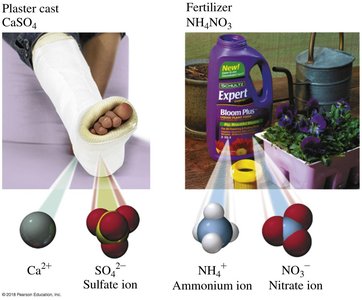
CaSO4 | Calcium sulfate | Plaster casts |
MgSO4 | Magnesium sulfate | Cathartic, Epsom salts |
K2CO3 | Potassium carbonate | Alkalizer, diuretic |
AgNO3 | Silver nitrate | Topical anti-infective |
NaHCO3 | Sodium bicarbonate | Antacid |
Zn3(PO4)2 | Zinc phosphate | Dental cement |
Lewis Structures and the Octet Rule
Lewis structures are used to represent the arrangement of electrons in molecules. The octet rule states that atoms tend to have eight electrons in their valence shell, except for certain exceptions.
Octet Rule: Most atoms achieve stability with eight valence electrons.
Exceptions: H2 (2 electrons), BCl3 (6 electrons), PCl5 (10 electrons), SF6 (12 electrons), ClO4 (14 electrons).
Naming Molecular Compounds
Molecular compounds are named using prefixes to indicate the number of atoms. If the first element is 'mono', the prefix is omitted.
Prefixes: mono-, di-, tri-, tetra-, penta-, hexa-, etc.
Example: CO2 is carbon dioxide, not monocarbon dioxide.
Atomic Size and Electronegativity Trends
Atomic size and electronegativity are periodic trends that influence chemical bonding.
Atomic Size: Increases down a group, decreases across a period.
Electronegativity: Increases across a period, decreases down a group.
Bond Polarity and Dipoles
The difference in electronegativity between atoms determines bond polarity. If the difference is 0.5 or greater, the bond is polar; otherwise, it is nonpolar.
Bond Polarity: Polar bonds have unequal sharing of electrons, creating dipoles.
Dipole Moment: A measure of molecular polarity.
Valence Shell Electron Pair Repulsion (VSEPR) Theory
VSEPR theory explains the geometry and polarity of covalent compounds. Electron groups (lone pairs or bonded atoms) arrange themselves to minimize repulsion.
Electron Groups: Lone pairs and bonded atoms.
Molecular Shape: Determined by the arrangement of electron groups.
Polarity: Determined by the sum of bond dipoles; equal and opposite dipoles cancel.
Molecular Shapes
Molecules can have various shapes depending on the number of electron groups and lone pairs.
Linear: 2 electron groups, 180° bond angle.
Trigonal Planar: 3 electron groups, 120° bond angle.
Bent: 3 electron groups with a lone pair.
Tetrahedral: 4 electron groups, 109.5° bond angle.
Trigonal Pyramidal: 4 electron groups with a lone pair.
Intermolecular Forces
Intermolecular forces are attractions between molecules, affecting physical properties like melting and boiling points.
Dipole-Dipole: Occurs in polar molecules.
Hydrogen Bonding: Extreme dipole-dipole, only for N, O, and F.
Dispersion (London) Forces: Weakest, present in all molecules, strength increases with size.
Melting and Boiling Points
The strength of intermolecular forces determines melting and boiling points.
Ionic Forces: Strongest, highest melting and boiling points.
Dipole-Dipole: Intermediate strength.
Dispersion Forces: Weakest, lowest melting and boiling points.
Example: Mg2+ has stronger attraction than Na+; O-H hydrogen bond is stronger than N-H due to greater electronegativity difference; bromine has stronger forces than chlorine due to larger atomic radius.
