 Back
BackLipids: Structure, Classification, and Biological Functions
Study Guide - Smart Notes

Lipids: Structure and Classification
Overview of Lipids
Lipids are a diverse group of biomolecules that play essential roles in biological systems, including energy storage, membrane structure, and signaling. They are characterized by their solubility in organic solvents and insolubility in water.
Key Feature: Lipids contain fatty acids or a steroid nucleus.
Biological Importance: Lipids are crucial in cell membranes, fat-soluble vitamins, and steroid hormones.
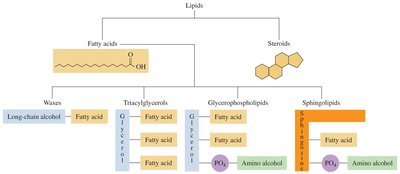
Types of Lipids
Lipids are classified based on their structure and hydrolysis properties:
Esters: Lipids that can be hydrolyzed to yield fatty acids and other molecules (e.g., waxes, fats, oils, phospholipids).
Steroids: Lipids that do not contain fatty acids and cannot be hydrolyzed; characterized by a four-ring steroid nucleus.
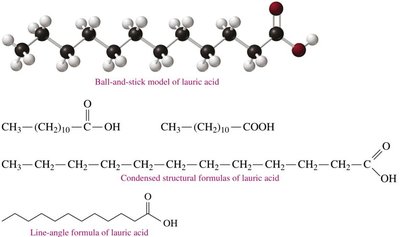
Fatty Acids: Structure and Classification
Structural Representation of Fatty Acids
Fatty acids are long hydrocarbon chains with a terminal carboxylic acid group. They can be represented in several ways:
Ball-and-stick model
Condensed structural formula
Line-angle formula
Saturated vs. Unsaturated Fatty Acids
Fatty acids are classified based on the presence of double bonds:
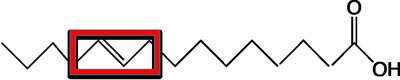
Saturated fatty acids: Only C–C single bonds; higher melting points; usually solid at room temperature.
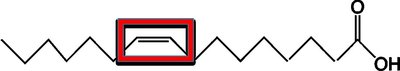
Unsaturated fatty acids: Contain C=C double bonds; lower melting points; usually liquid at room temperature.
Monounsaturated: One C=C double bond.
Polyunsaturated: Two or more C=C double bonds.

Examples of Fatty Acids
Lauric acid: Saturated, 12 carbons, found in coconut oil.
Oleic acid: Monounsaturated, 18 carbons, found in olive oil.
Linoleic acid: Polyunsaturated, 18 carbons, found in sunflower oil.
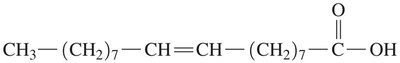
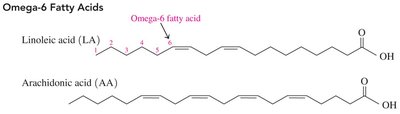
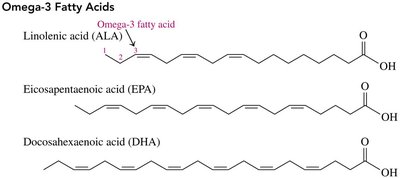
Omega Fatty Acids
Omega-6 fatty acids: First double bond at carbon 6 from the methyl end (e.g., linoleic acid, arachidonic acid).
Omega-3 fatty acids: First double bond at carbon 3 from the methyl end (e.g., linolenic acid, EPA, DHA).
Waxes and Triglycerols
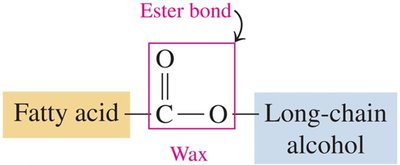
Waxes
Waxes are esters formed from a fatty acid and a long-chain alcohol. They serve as protective coatings in plants and animals.
Structure: Ester bond between fatty acid and alcohol.
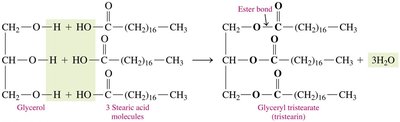
Triglycerols (Triacylglycerols)
Triglycerols are formed by esterification of glycerol with three fatty acids. They are the main form of energy storage in animals.
Formation: Glycerol + 3 fatty acids → triglycerol + 3 H2O
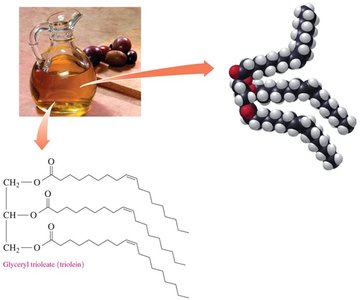
Energy Storage and Fatty Acid Composition
Animal fats: Higher percentage of saturated fatty acids; solid at room temperature.
Vegetable oils: Higher percentage of unsaturated fatty acids; liquid at room temperature.
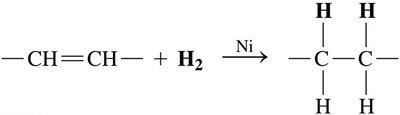
Chemical Reactions of Lipids
Hydrogenation
Hydrogenation converts unsaturated fats to saturated fats by adding hydrogen to double bonds, often producing trans fats.
Reaction:
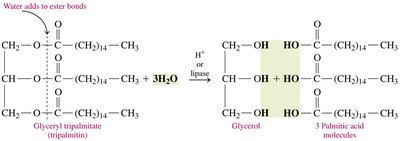
Hydrolysis
Hydrolysis splits triglycerols into glycerol and fatty acids, catalyzed by acids or enzymes (lipases).
Reaction: Triglycerol + 3 H2O → glycerol + 3 fatty acids
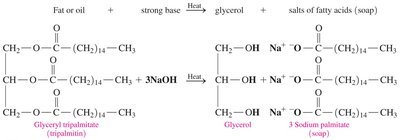
Saponification
Saponification is the reaction of a fat with a strong base to produce glycerol and soap (salts of fatty acids).
Reaction: Triglycerol + 3 NaOH → glycerol + 3 sodium salts of fatty acids
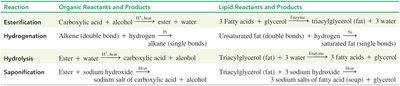
Summary Table of Lipid Reactions
Reaction | Organic Reactants and Products | Lipid Reactants and Products |
|---|---|---|
Esterification | Carboxylic acid + alcohol → ester + water | 3 fatty acids + glycerol → triglycerol + 3 water |
Hydrogenation | Alkene + hydrogen → alkane | Unsaturated fat + hydrogen → saturated fat |
Hydrolysis | Ester + water → carboxylic acid + alcohol | Triglycerol + 3 water → 3 fatty acids + glycerol |
Saponification | Ester + sodium hydroxide → sodium salt + alcohol | Triglycerol + 3 sodium hydroxide → 3 sodium salts + glycerol |
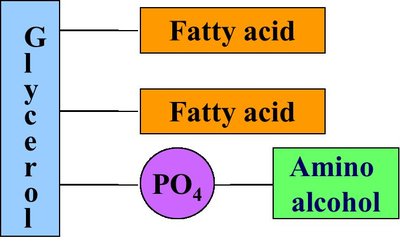
Phospholipids and Cell Membranes
Phospholipid Structure
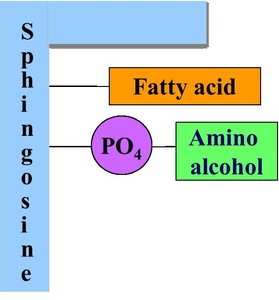
Phospholipids are major components of cell membranes, consisting of a glycerol or sphingosine backbone, two fatty acids, a phosphate group, and an amino alcohol.
Glycerophospholipids: Glycerol + 2 fatty acids + phosphate + amino alcohol
Sphingomyelins: Sphingosine + fatty acid + phosphate + amino alcohol
Lecithin and Cephalin
Lecithin and cephalin are types of glycerophospholipids found in brain and nerve tissues, egg yolk, wheat germ, and yeast. 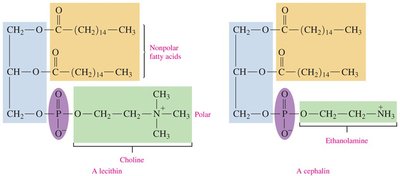
Cell Membrane Structure
Cell membranes are composed of a lipid bilayer with embedded proteins and cholesterol, providing fluidity and selective permeability.
Fluid Mosaic Model: Proteins and cholesterol are embedded in a bilayer of phospholipids.
Transport: Diffusion, facilitated transport, and active transport mechanisms.
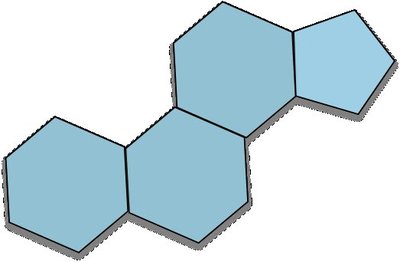
Steroids and Cholesterol
Steroid Structure
Steroids have a characteristic four-ring structure known as the steroid nucleus. 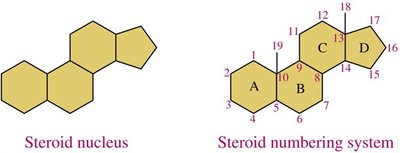
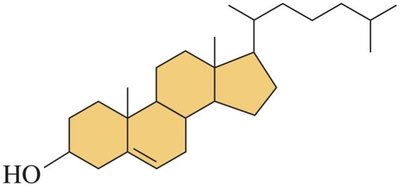
Cholesterol
Cholesterol is the most abundant steroid in the body, essential for cell membranes, brain and nerve tissue, steroid hormones, and vitamin D.
Structure: Contains a hydroxyl group, double bond, methyl groups, and an alkyl chain.
Health Impact: High levels can form plaques, leading to atherosclerosis.
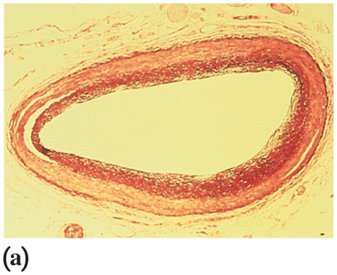
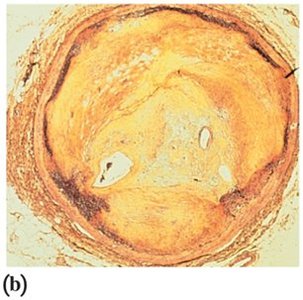
Lipoproteins
Lipoproteins transport nonpolar lipids and cholesterol in the bloodstream. Types include HDL (good cholesterol) and LDL (bad cholesterol). 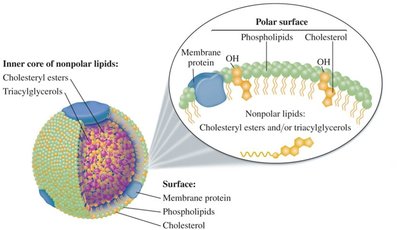
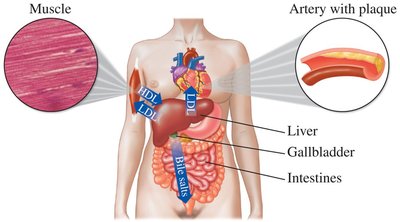
Steroid Hormones
Steroid hormones are chemical messengers produced from cholesterol, including testosterone, estrogens, progesterone, and adrenal corticosteroids. 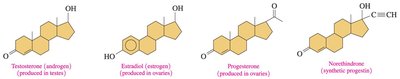
Summary
Lipids are essential biomolecules with diverse structures and functions, including energy storage, membrane structure, and signaling. Understanding their classification, chemical reactions, and biological roles is fundamental in biochemistry and health sciences.
