 Back
BackLipids: Structure, Properties, and Metabolism
Study Guide - Smart Notes

Lipids: Structure, Properties, and Metabolism
Introduction to Lipids
Lipids are a diverse group of naturally occurring molecules that are soluble in nonpolar organic solvents but insoluble in water. Their classification is based on physical properties rather than a specific chemical structure, which allows for a wide variety of functions in biological systems.
Functions of Lipids: Energy storage, structural components of cell membranes, and signaling molecules (e.g., steroids, eicosanoids).
General Structure: Most lipids contain hydrocarbon chains or rings, often with functional groups such as carboxylic acids, esters, or amides.
23.1 Structure and Classification of Lipids
Classification of Lipids
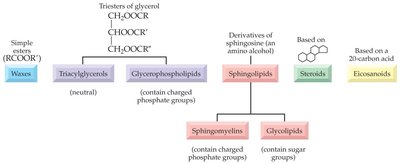
Lipids are classified based on their structure and function. The main categories include simple lipids, compound lipids, steroids, and miscellaneous lipids.
Simple Lipids: Esters of fatty acids with various alcohols (e.g., fats, oils, waxes).
Compound Lipids: Contain additional functional groups (e.g., phospholipids, glycolipids, sphingolipids).
Steroids: Lipids with a characteristic fused ring structure (e.g., cholesterol).
Eicosanoids: Lipids derived from 20-carbon fatty acids, involved in signaling.
23.2 Fatty Acids and Their Esters
Fatty Acids: Structure and Types
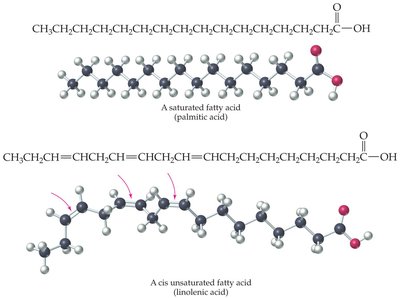
Fatty acids are long, unbranched hydrocarbon chains with a terminal carboxylic acid group. They are key building blocks of many lipids.
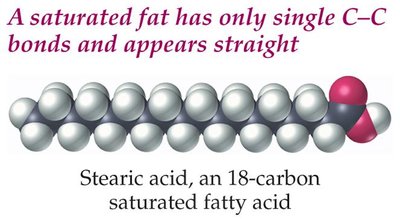
Saturated Fatty Acids: Contain only single bonds between carbon atoms (e.g., palmitic acid, stearic acid).
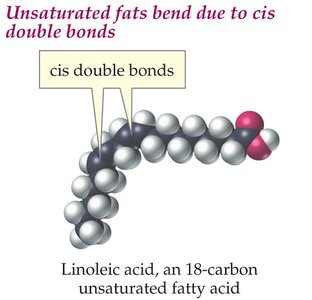
Unsaturated Fatty Acids: Contain one or more double bonds. Naturally occurring unsaturated fatty acids usually have cis double bonds, causing kinks in the chain (e.g., oleic acid, linoleic acid).
Essential Fatty Acids: Fatty acids that cannot be synthesized by the body and must be obtained from the diet (e.g., linoleic and linolenic acids).

Nomenclature of Fatty Acids
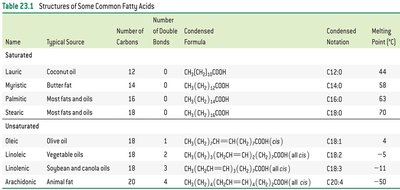
Shorthand Notation: C followed by the number of carbons, a colon, and the number of double bonds (e.g., C18:1 for oleic acid).
Omega (ω) System: Numbering starts from the methyl (tail) end; e.g., ω-3 fatty acids have a double bond three carbons from the tail.
Common Fatty Acids and Their Sources
Name | Number of Carbons | Number of Double Bonds | Source |
|---|---|---|---|
Lauric | 12 | 0 | Coconut oil |
Myristic | 14 | 0 | Butter fat |
Palmitic | 16 | 0 | Most fats and oils |
Stearic | 18 | 0 | Most fats and oils |
Oleic | 18 | 1 | Olive oil |
Linoleic | 18 | 2 | Vegetable oils |
Linolenic | 18 | 3 | Soybean and canola oils |
Arachidonic | 20 | 4 | Animal fat |
Fatty Acid Esters: Waxes, Fats, and Oils
Waxes: Esters of long-chain fatty acids with long-chain alcohols. Serve as protective coatings in plants and animals.
Fats and Oils: Triesters (triglycerides/triacylglycerols) formed from glycerol and three fatty acids. Fats are solid at room temperature (more saturated), oils are liquid (more unsaturated).

23.3 Properties of Fats and Oils
Physical Properties and Melting Points
The physical properties of fats and oils are determined by the degree of saturation and the length of the fatty acid chains.
Saturated Fats: Straight chains pack closely, resulting in higher melting points (solid at room temperature).
Unsaturated Fats: Cis double bonds introduce kinks, preventing tight packing and lowering melting points (liquid at room temperature).
Examples of Fats and Oils
Animal Fats: High in saturated fatty acids (e.g., butter, lard).
Vegetable Oils: High in unsaturated fatty acids (e.g., olive oil, soybean oil).


23.4 Chemical Reactions of Triacylglycerols
Hydrogenation
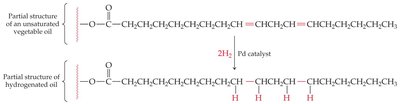
Hydrogenation is the process of adding hydrogen to unsaturated fatty acids, converting double bonds to single bonds and producing saturated fats. Partial hydrogenation can create trans fats, which are associated with negative health effects.
Equation:
Trans Fats: Formed during partial hydrogenation; have straighter chains and higher melting points than cis fats.
Hydrolysis and Saponification
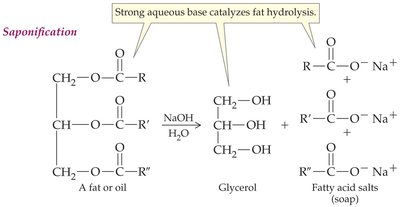
Triacylglycerols can be hydrolyzed by acids, bases, or enzymes. Base-catalyzed hydrolysis (saponification) produces glycerol and soap (salts of fatty acids).
Equation:
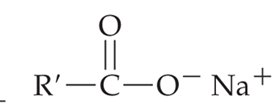
Soap: Amphiphilic molecules that form micelles, allowing grease and oils to be suspended in water and rinsed away.
24.1 Digestion of Triacylglycerols
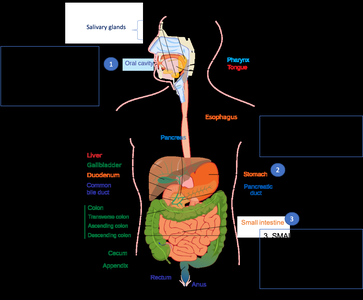
Digestion and Absorption
Dietary triacylglycerols are digested in the small intestine with the help of bile acids and pancreatic lipases. The products are absorbed and transported in the blood as lipoproteins.
Bile Acids: Emulsify fats, forming micelles for enzyme action.
Lipoproteins: Chylomicrons transport dietary lipids through the lymphatic system and bloodstream.
24.2 Lipoproteins for Lipid Transport
Major Classes of Lipoproteins
Lipoproteins are complexes that transport lipids in the aqueous environment of blood. They are classified by density, which depends on the ratio of lipid to protein.
Type | Density (g/cm3) | Main Function |
|---|---|---|
Chylomicrons | <0.95 | Transport dietary triacylglycerols to tissues |
VLDL | 0.96–1.006 | Transport triacylglycerols from liver to tissues |
IDL | 1.007–1.019 | Carry VLDL remnants back to liver |
LDL | 1.020–1.062 | Transport cholesterol to tissues |
HDL | 1.063–1.210 | Transport cholesterol from tissues to liver |
24.3 Triacylglycerol Metabolism: Storage and Mobilization
Storage and Mobilization of Triacylglycerols
Triacylglycerols are stored in adipose tissue as energy reserves. Their synthesis and breakdown are regulated by hormones such as insulin and glucagon.
Synthesis: Glycerol 3-phosphate and fatty acyl-CoA are combined to form triacylglycerols.
Mobilization: When energy is needed, triacylglycerols are hydrolyzed to release fatty acids and glycerol into the bloodstream.
24.5 Oxidation of Fatty Acids (β-Oxidation)
β-Oxidation Pathway
Fatty acid oxidation occurs in the mitochondria and involves the sequential removal of two-carbon units as acetyl-CoA. This process generates NADH and FADH2 for ATP production.
Steps:
Activation: Fatty acid + CoA + ATP → Fatty acyl-CoA
Transport: Fatty acyl-CoA enters mitochondria via carnitine shuttle
β-Oxidation Spiral: Four recurring reactions remove two-carbon acetyl-CoA units
Energy Yield: Fatty acids yield more ATP per gram than carbohydrates due to their higher degree of reduction.
Example Calculation: Complete oxidation of stearic acid (C18) yields significantly more ATP than glucose of equivalent carbon number.
24.6 Ketone Bodies and Ketoacidosis
Ketone Body Formation
When fatty acid oxidation produces more acetyl-CoA than the citric acid cycle can process, excess acetyl-CoA is converted into ketone bodies (acetoacetate, β-hydroxybutyrate, acetone) in the liver.
Physiological Role: Ketone bodies serve as alternative energy sources during fasting, starvation, or uncontrolled diabetes.
Ketoacidosis: Excessive ketone body production lowers blood pH, leading to dangerous metabolic acidosis.
24.7 Biosynthesis of Fatty Acids (Lipogenesis)
Fatty Acid Synthesis
Lipogenesis is the anabolic pathway for synthesizing fatty acids from acetyl-CoA, primarily in the cytosol of liver and adipose cells. The process is not a simple reversal of β-oxidation and involves distinct enzymes and intermediates.
Key Steps:
Acetyl-CoA is converted to malonyl-CoA (rate-limiting step).
Fatty acid synthase complex elongates the chain two carbons at a time, using malonyl-CoA as the donor.
Palmitic acid (C16:0) is the primary product, which can be further elongated or desaturated.
Integration with Metabolism: Excess carbohydrates and proteins can be converted to fatty acids for long-term energy storage.
Summary Table: Comparison of Fats and Oils
Property | Fats | Oils |
|---|---|---|
Physical State (Room Temp) | Solid | Liquid |
Saturation | Mostly saturated | Mostly unsaturated |
Source | Animal | Plant |
Melting Point | Higher | Lower |
Worked Example: Digestion and Absorption of Dietary Fat
As dietary fat (e.g., from ice cream) is consumed, it is emulsified by bile acids in the small intestine, hydrolyzed by pancreatic lipases to mono- and diacylglycerols and fatty acids, absorbed by intestinal cells, reassembled into triacylglycerols, and transported in chylomicrons to the liver for processing.
