 Back
BackMatter and Energy: Classification, Properties, and Changes
Study Guide - Smart Notes

Ch. 3: Matter and Energy
Chemical Change vs. Physical Change
Understanding the distinction between chemical and physical changes is fundamental in chemistry. Chemical changes result in the formation of new substances, while physical changes alter the form or appearance without changing the substance's identity.
Chemical Change: A process in which a substance is transformed into a new substance, changing its identity. Evidence includes color change, gas production, formation of a precipitate, or energy changes (heat/light).
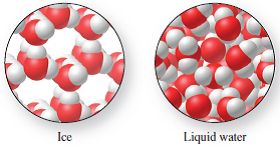
Physical Change: The identity of the substance remains the same, but its physical form changes (e.g., melting, boiling, dissolving). Evidence includes changes in state, shape, or size.
Example: Burning magnesium in acid is a chemical change; melting ice is a physical change.
Mixtures and Separating Mixtures
Mixtures consist of two or more substances physically combined. The components of mixtures can be separated by physical means without altering their chemical identities.
Mixture: A combination of two or more substances where each retains its own properties.
Physical Separation: Methods include filtration, magnetism, and decanting.
Example: Iron filings can be separated from sulfur using a magnet.




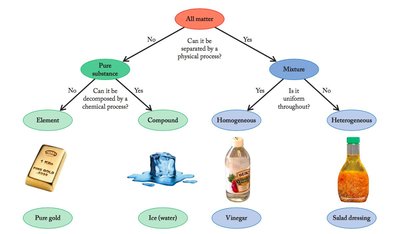
Classifying Matter
Matter can be classified as pure substances or mixtures. Pure substances include elements and compounds, while mixtures can be homogeneous or heterogeneous.
Pure Substance: Has a uniform and definite composition. Includes elements and compounds.
Element: A substance made of only one type of atom (e.g., gold).
Compound: A substance composed of two or more elements chemically bonded in a fixed ratio (e.g., water).
Mixture: Can be separated by physical processes. Homogeneous mixtures are uniform throughout (e.g., vinegar), while heterogeneous mixtures are not (e.g., salad dressing).















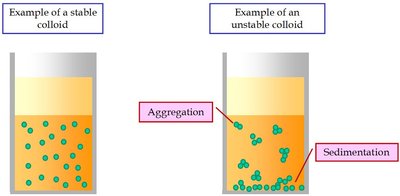

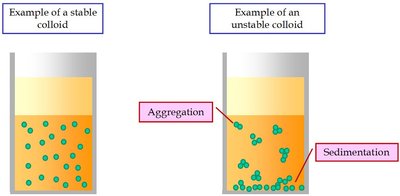



Physical and Chemical Changes: Practice
Classifying changes as physical or chemical is a key skill in chemistry. Practice problems help reinforce this understanding.
Physical Change: Example: Ice melting, mixing chocolate syrup with milk.
Chemical Change: Example: Coal burning, explosion of a firecracker.
Temperature and Temperature Scales
Temperature is a measure of the average kinetic energy of particles. Common scales include Celsius, Fahrenheit, and Kelvin.
Celsius (°C): Water freezes at 0°C and boils at 100°C.
Fahrenheit (°F): Water freezes at 32°F and boils at 212°F.
Kelvin (K): Absolute temperature scale; 0 K is absolute zero.
Conversion formulas:
Energy: Definitions and Units
Energy is the capacity to supply heat or do work. It is measured in various units, including joules, calories, and kilowatt-hours.
Joule (J): SI unit of energy.
Calorie (cal): Amount of energy needed to raise 1 g of water by 1°C.
British Thermal Unit (BTU): Used in heating and cooling systems.
Energy Conversion Table:
Unit | Equivalent |
|---|---|
1 joule | 1 kg·m2/s2 |
1 calorie | 4.184 J |
1,000 calories | 1 kcal = 1 Calorie |
1 BTU | 1,055 J |
1 kWh | 3.6 × 106 J |


Kinetic and Potential Energy
Energy exists in two main forms: kinetic (energy of motion) and potential (stored energy).
Kinetic Energy: Energy due to motion.
Potential Energy: Energy stored due to position or arrangement.
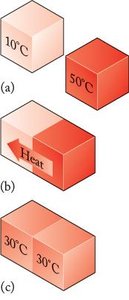
Heat (q): Energy transfer due to temperature difference.
Work (w): Energy transfer due to force acting over a distance.
Formula for heat:
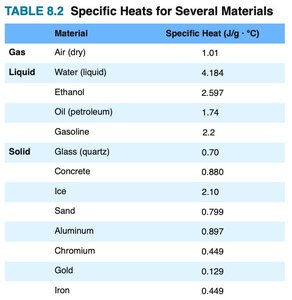
Specific Heat
Specific heat is the amount of heat required to raise the temperature of 1 gram of a substance by 1°C. Different materials have different specific heats.
Formula:
Example: Water has a high specific heat (4.184 J/g·°C), making it effective for temperature regulation.
Material | Specific Heat (J/g·°C) |
|---|---|
Air (dry) | 1.01 |
Water (liquid) | 4.184 |
Ice | 2.10 |
Aluminum | 0.897 |
Gold | 0.129 |
Iron | 0.449 |


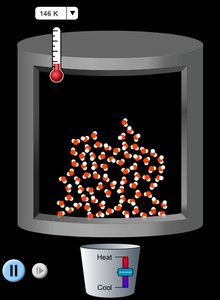
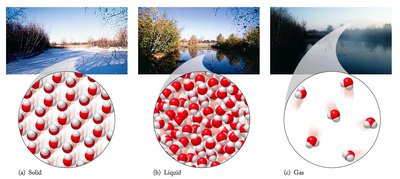
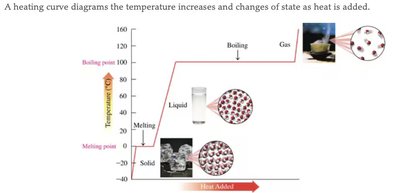
States of Matter and Changes of State
Matter exists in three primary states: solid, liquid, and gas. Each state has distinct properties, and changes of state occur with energy transfer.
Solid: Definite shape and volume.
Liquid: Definite volume, no definite shape.
Gas: Neither definite shape nor volume.
Changes of State: Include melting, freezing, boiling, condensation, and sublimation.
Heating Curve: Shows temperature changes and phase transitions as heat is added.

Additional info: Practice problems and Kahoot! exercises are included to reinforce classification and calculation skills. These notes cover all major aspects of Ch. 3: Matter and Energy, including classification, properties, changes, and energy concepts.
